Pharmaceutical Microbiology Market Outlook:
Pharmaceutical Microbiology Market size was over USD 7.03 billion in 2025 and is anticipated to cross USD 16.8 billion by 2035, witnessing more than 9.1% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of pharmaceutical microbiology is assessed at USD 7.61 billion.

The market is majorly witnessing growth due to the increase in the occurence of infectious disorders. Infectious diseases like HIV, sexually transmitted infections (STDs), and other infectious disorders including hepatitis A, B, and C are becoming more common across the globe. For instance, worldwide around 39 million persons were found to be HIV positive in 2022. The market is thus driven by the growing prevalence of infectious illness cases caused by contaminated needle injections, homelessness, unprotected sex, lack of access to healthcare, and several other socioeconomic difficulties.
In addition, regulatory agencies such as the FDA and EMA, have stringent guidelines mandating the sterility of pharmaceutical products to ensure patient safety. Compliance with these regulations necessitates throughout the manufacturing process, driving the demand for microbiology services and products. Also, maintaining sterility is essential for preserving the efficacy and safety of pharmaceutical products. Therefore, these factors are accelerating the growth of the market.
Key Pharmaceutical Microbiology Market Insights Summary:
Regional Highlights:
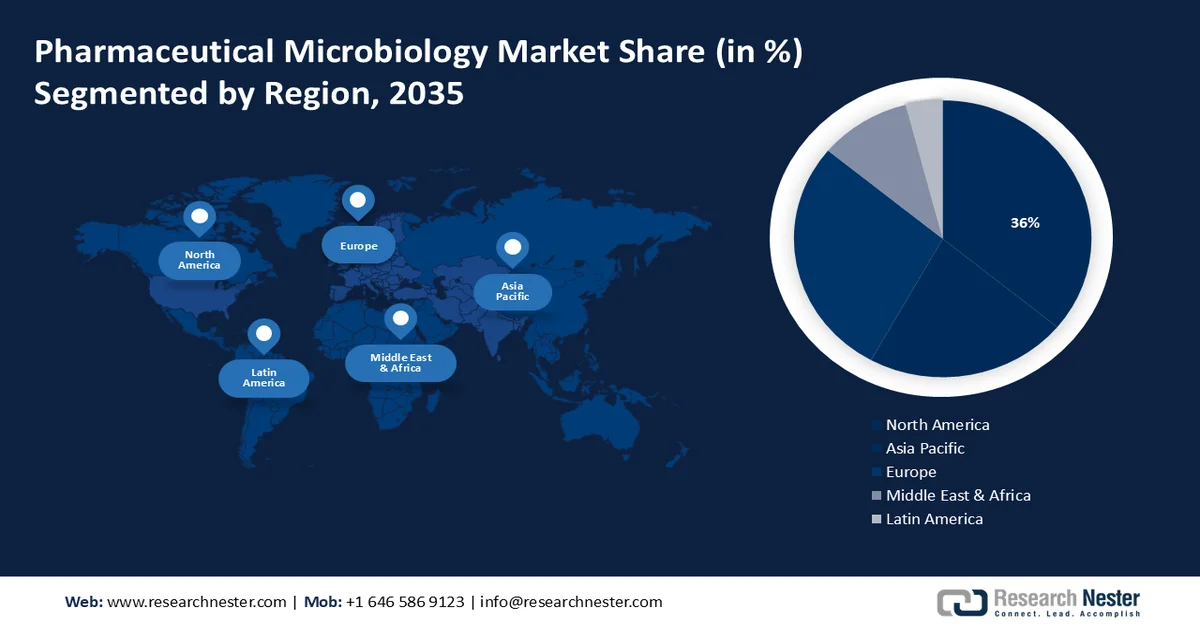
- North America in the pharmaceutical microbiology market is estimated to hold the largest revenue share of 36% by 2035, driven by growing innovation in testing technologies.
- Asia Pacific region is projected to capture around 28% revenue share by 2035, impelled by rising investments in biotechnology and increasing demand for biologics.
Segment Insights:
- Biopharmaceutical segment in the pharmaceutical microbiology market is projected to account for over 54% share by 2035, owing to the rising demand for biologic pharmaceuticals among aging populations.
- Microbial identification segment is expected to hold more than 35% revenue share by 2035, fueled by increasing emphasis on contamination detection and rapid microbiological techniques.
Key Growth Trends:
- Rising investment in the healthcare infrastructure
- Growing demand for sterility of the products
Major Challenges:
- Limited shelf life of microorganisms and microbiology reagants
- Emergence of new microbial pathogens and drug-resistant strains may hinder the growth of the market
Key Players: Siemens, Schneider Electric, Danfoss, Honeywell International Inc., IMI plc, Sauter AG, Emmeti, REHAU Group, Uponor, Belimo Holding AG.
Global Pharmaceutical Microbiology Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 7.03 billion
- 2026 Market Size: USD 7.61 billion
- Projected Market Size: USD 16.8 billion by 2035
- Growth Forecasts: 9.1% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (36% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, China, Japan, United Kingdom
- Emerging Countries: China, India, Japan, South Korea, Brazil
Last updated on : 25 February, 2026
Pharmaceutical Microbiology Market - Growth Drivers and Challenges
Growth Drivers
- Rising investment in the healthcare infrastructure - With the expanding healthcare infrastructure, more people gain access to healthcare services which leads to growing demand for pharmaceutical products. Also, investment in healthcare infrastructure usually aims to address public health issues, such as infectious diseases and antimicrobial resistance. For example, the 2023 report provides recent insights into the development of world’s health spending over the peak of the COVID-19 pandemic. The analysis that was released in advance of Universal Health Coverage (UHC) Day, shows that, in 2021, health spending globally reached a record high of USD 9.8 trillion, or 10.3% of the world's gross domestic product (GDP). Nevertheless, there was an apparent discrepancy in the allocation of spending. The pharmaceutical microbiology market is expanding because public health spending has gone up globally.
- Growing demand for sterility of the products - Sterile testing is required to guarantee the safety and effectiveness of biopharmaceuticals, such as cell treatments, monoclonal antibodies, and vaccines, due to the increasing demand for these products. For instance, the stem cell therapy industry was estimated to be worth approximately USD 755 million globally in 2018, and it is expected to grow to over 11 billion dollars by 2029.
To avoid pharmaceutical items from harming patients and causing infections and other health issues, sterility is essential. Additionally, keeping pharmaceutical products sterile is crucial to preserving their quality and integrity over the course of their shelf life and guaranteeing that they are safe and effective for use by patients. - Growing technological advancements - The rapid on-site identification of microbiological contaminations made possible by advancements in point-of-care testing technologies allows for prompt decision-making and intervention to prevent contamination in pharmaceutical manufacturing facilities. Furthermore, high-throughput screening of microbiological samples with low reagent usage is made possible by miniaturized platforms and microfluidic devices, which makes them perfect for large-scale testing in pharmaceutical labs.
Additionally, real-time monitoring, trend analysis, and predictive modeling of the dangers associated with microbial contamination are made possible by the integration of microbiological technologies with data analytics and artificial intelligence, which is propelling the pharmaceutical microbiology market's expansion.
Challenges
- Limited shelf life of microorganisms and microbiology reagants - The duration of microbiology reagents' shelf life is contingent upon their exposure to ambient conditions. Because microorganisms, like bacteria, have a short lifespan (12 hours), there is very little time for microbiological study because the microorganisms' quality deteriorates and they become inappropriate for use, leading to erroneous results from pharma microbiology testing kits. Thus, the limited or brief shelf life of microbiological reagents may result in higher expenses and hamper the growth of the market.
- Emergence of new microbial pathogens and drug-resistant strains may hinder the growth of the market
- Complexity of manufacturing processes of the biologics and advanced therapies which may act as a hindrance to the market growth.
Pharmaceutical Microbiology Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
9.1% |
|
Base Year Market Size (2025) |
USD 7.03 billion |
|
Forecast Year Market Size (2035) |
USD 16.8 billion |
|
Regional Scope |
|
Pharmaceutical Microbiology Market Segmentation:
Product Type Segment Analysis
In pharmaceutical microbiology market, biopharmaceutical segment is likely to capture over 54% share by 2035. Biopharmaceuticals have a high therapeutic value and can successfully treat a wide range of chronic ailments, such as cancer, diabetes, cardiovascular disease, Parkinson's disease, and many more since they are produced utilizing living bacteria and animal cells. Furthermore, it is anticipated that the need for biologic pharmaceuticals would increase as the global population ages and becomes more vulnerable to chronic disorders.
Pharmaceutical companies are also making significant investments in the development of biological medications, which is creating a growing pipeline of cutting-edge treatments that address a range of illnesses. For example, Novartis's Cosentyx® (secukinumab) has been licensed by the US Food and Drug Administration (FDA) to treat people with moderate to severe hidradenitis suppurative (HS). Cosentyx.2 is the sole FDA-licensed completely human biopharmaceutical that directly suppresses interleukin-17A (IL-17A), a cytokine believed to be involved in HS inflammation.
Service Segment Analysis
In pharmaceutical microbiology market, microbial identification segment is predicted to hold more than 35% revenue share by 2035. With the expanding complexity of pharmaceutical manufacturing processes and the worldwide nature of supply chains, pharmaceutical companies are increasingly concerned about microbial contamination. As a result, there is a growing emphasis on microbiological identification to detect and treat contamination issues promptly, ensuring product quality and safety. Furthermore, technological advancements have resulted in the development and acceptance of rapid microbiological techniques (RMM) for microbial identification, which is expected to rise further in the future.
Also, the advent of new microbial diseases is also propelling the growth of the segment. Some microbial diseases include tuberculosis, Lyme disease, measles, malaria, fungal diseases, and others. According to the most recent estimates, there are 476,000 new Lyme cases in the United States each year, and is expected to increase by 59% by 2021. Scientists predict that two million people will suffer from post-treatment Lyme disease by the end of 2020. Microbial identification is critical for tracking and characterizing these infections to establish effective control measures and treatment plans.
Our in-depth analysis of the global market includes the following segments:
|
Type |
|
|
Tests |
|
|
End-user |
|
|
Services |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Pharmaceutical Microbiology Market - Regional Analysis
North American Market Insights
North America industry is estimated to hold largest revenue share of 36% by 2035, due to growing innovation in testing technologies in the region. Due to its better health care system and stringent regulations, the region has a higher demand for pharmaceutical microbiology products. Moreover, innovation in testing technologies is encouraged by the existence of large research and development, biotechnology, and pharmaceutical companies.
Other factors contributing to the growth of the market in this region include an aging population, as well as increasing incidences of infectious diseases in the region such as HIV and Influenza. For instance, in the US approximately 25,000 people died from the disease during the flu season in 2019-2020.
APAC Market Insights
By the end of 2035, Asia Pacific region in pharmaceutical microbiology market is set to dominate around 28% revenue share. The rapid growth of regional nations has led to grown spending on health care, which is driving the demand for pharmaceutical products & services. In addition, the market demand in the region is driven by growing investments in biotechnology and increasing demand for biologics. Moreover, factors such as the availability of skilled labor and supportive regulatory environments have made this region a key hub for pharmaceutical manufacturing activities.
Key Pharmaceutical Microbiology Market Players:
- NEOGEN Corporation
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Hardy Diagnostics
- IDEXX
- Charles River Laboratories
- BioMerieux SA
- Merck KGAA
- Rapid Micro Biosystems.
- Abbott Laboratories, Inc
- Bio-Rad Laboratories
- Gen-Probe, Inc
Recent Developments
- NEOGEN Corporation unveiled Soleris® Next Generation (NG). The novel Soleris® Next Generation (NG) from NEOGEN is capable of quickly identifying a broad range of bacteria that may jeopardize the quality and safety of food and other consumer goods. For instance, whereas traditional methods require three to five days to identify and count yeast and mold in a sample, Solaris NG can do so in just 48 hours. Additionally, it can identify Alicyclobacillus, a bacterium of special concern to the fruit juice industry, in just 48 hours as opposed to five days using other methods.
- Hardyc Diagnostics designed CmpactDry TCR, a ready-to-use dehydrated culture medium plate to identify and count microorganisms. It can yield total aerobic plate counts in as little as 24 hours. Being AOAC PTM (Association of Official Analytical Chemists - Performance Tested Methods) approved, this self-diffusing plate is the simplest to use for plate counts since it doesn't require spreading or stamping. Just take off the lid, pour 1 milliliter of sample onto the middle of the plate, replace the lid, and let it incubate.
- Report ID: 5870
- Published Date: Feb 25, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Pharmaceutical Microbiology Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




