Peripheral Vascular Devices Market Outlook:
Peripheral Vascular Devices Market size was valued at over USD 10.1 billion in 2025 and is expected to reach USD 17.3 billion by the end of 2035, growing at a CAGR of 6.2% during the forecast period, i.e., 2026-2035. In 2026, the industry size of peripheral vascular devices is estimated at USD 10.7 billion.

The worldwide peripheral vascular devices market is continuously undergoing a transformative phase, which is characterized by increased technological innovation, shifting care settings, evolving demographic patterns, a surge in prioritizing minimally invasive interventions, and a rise in chronic diseases. According to official statistics published by NLM in January 2025, a clinical study was conducted on 408 hospitals regarding minimally invasive technologies. Of these hospitals, 153, which is 38%, have significantly unveiled robotic-assisted surgery (RAS), which is extremely suitable for conducting common general surgeries. In addition, the relatively minimally invasive surgery for hospitals that introduced the RAS was 1.08, in comparison to hospitals that did not adopt the technique, along with an index rate of 1.1, thereby making it suitable for boosting the peripheral vascular devices market expansion across different countries.
Furthermore, the shift towards ambulatory surgical centers and office-based laboratories, the integration of therapeutic and imaging devices, an expansion in drug-eluting technological platforms, a rise in specialized devices for calcified lesion treatment, and geographic expansion into emerging economies are certain trends that are proliferating the market growth. As stated in an article published by NLM in January 2025, the payer’s pricing of a remote patient monitoring for hypertension (RPM-HTN) program amounted to USD 330 per patient, while usually ranging between USD 208 and USD 452, along with an annual program expense of USD 33,000 for 100 patients globally. Besides, notable expenses for such treatments include USD 172 per patient for data evaluation by nurses, USD 48 for blood pressure device costs, and USD 36 per patient for nurse and patient communication, thus enhancing the peripheral vascular devices market exposure.
Key Peripheral Vascular Devices Market Insights Summary:
Regional Highlights:
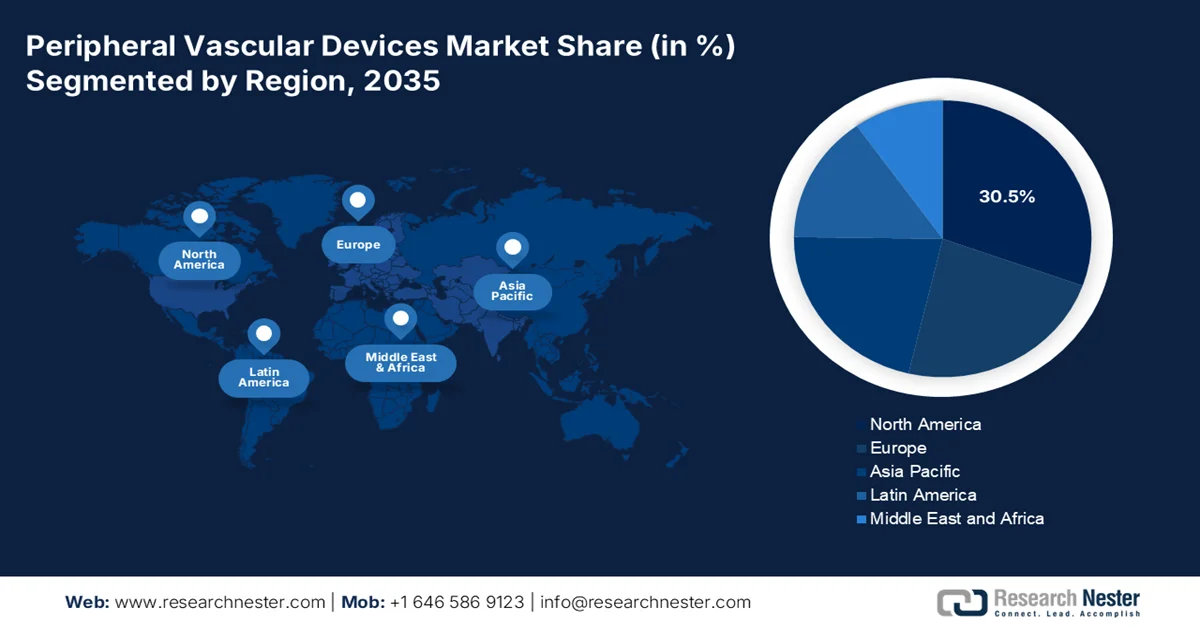
- North America peripheral vascular devices market is projected to capture a 30.5% share by 2035, impelled by rising prevalence of peripheral artery disease supported by aging demographics, obesity, and diabetes incidence
- Asia Pacific is expected to register the fastest growth over the forecast period, stimulated by increasing lifestyle-related conditions, expanding elderly population, and growing adoption of minimally invasive procedures
Segment Insights:
- Hospitals segment in the peripheral vascular devices market is anticipated to account for an 85.2% share by 2035, driven by their capability to perform complex vascular interventions supported by advanced imaging infrastructure and multidisciplinary expertise
- Iliac artery sub-segment is projected to secure the second-largest share during the forecast period, fueled by its critical anatomical role in supplying blood to lower limbs and pelvic organs along with structural variability supporting procedural demand
Key Growth Trends:
- Rise in Vascular complications
- Demographic aging across developed economies
Major Challenges:
- Regulatory complexity and post-industry safety challenges
- Economic barriers across public healthcare systems
Key Players: Medtronic (Ireland), Boston Scientific Corporation (U.S.), Abbott Laboratories (U.S.), B. Braun SE (Germany), W. L. Gore & Associates, Inc. (U.S.), Edwards Lifesciences Corporation (U.S.), Terumo Corporation (Japan), Stryker Corporation (U.S.), BD (Becton, Dickinson and Company) (U.S.), Cook Group (U.S.), AngioDynamics, Inc. (U.S.), Cardiovascular Systems, Inc. (U.S.), LeMaitre Vascular, Inc. (U.S.), BIOTRONIK (Germany), JOTEC GmbH (Germany), Braile Biomédica (Brazil), Control Medical Technology, LLC (U.S.), Walk Vascular, LLC (U.S.), Transit Scientific (U.S.), Conic Vascular (U.S.), Siemens Healthineers (Germany).
Global Peripheral Vascular Devices Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 10.1 billion
- 2026 Market Size: USD 10.7 billion
- Projected Market Size: USD 17.3 billion by 2035
- Growth Forecasts: 6.2% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (30.5% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, China, United Kingdom
- Emerging Countries: India, South Korea, Brazil, Mexico, Malaysia
Last updated on : 30 March, 2026
Peripheral Vascular Devices Market - Growth Drivers and Challenges
Growth Drivers
- Rise in Vascular complications: The accelerating worldwide burden of diabetes mellitus readily serves as one of the primary drivers for the peripheral vascular devices market. According to official statistics published by NLM in March 2024, environmental and hereditary factors significantly contribute to the etiopathogenesis of type 2 diabetes, which effectively makes up more than 90% of overall incidences. Based on this, it has been estimated that 240 million patients reside with undiagnosed diabetes, effectively imposing aggressive financial strain on global healthcare systems. In addition, approximately 537 million, which is 10.5% of individuals aged between 20 and 79 years, are presently managing the disease. Besides, as per the International Diabetes Federation (IDF) estimates that the global healthcare costs amount to USD 966 billion, thus denoting a huge growth opportunity for the peripheral vascular devices market.
- Demographic aging across developed economies: The aspect of population aging constitutes a fundamental driver for the peripheral vascular devices market, since the disease is increasingly prevalent with advancing age. As stated in an article published by the World Health Organization (WHO) in October 2025, by the end of 2030, 1 in 6 people across different regions is predicted to be aged more than 60 years. In this regard, the population share in this particular age category is poised to increase from 1 billion to 1.4 billion. Besides, by the end of 2050, the world’s elderly population is also expected to double accounting for 2.1 billion, and meanwhile, the number of persons aged more than 80 years is predicted to triple and reach 426 million, thus positively impacting the market development.
- Expansion of healthcare infrastructure: The continuous technological advancement in the peripheral vascular devices market has made expansions in treatment indications that optimize clinical results and ensure intervention among the patient population. As stated in an article published by NLM in February 2025, peripheral artery disease is one of the most underdiagnosed and prevalent atherosclerotic conditions that affects more than 10 million adults, especially in the U.S. Therefore, to keep control of the disease prevalence, the PAD screening technique takes into consideration routine screening that includes major medical surgeries that are involved in managing the disease. This particular technique is suitable for patients aged between 50 and 65 years, thereby denoting an optimistic outlook for the market globally.
Challenges
- Regulatory complexity and post-industry safety challenges: The regulatory landscape for the peripheral vascular devices market presents formidable barriers to market entry and sustained commercialization. The U.S. Food and Drug Administration (FDA) mandates rigorous premarket approval processes for high-risk devices, requiring substantial clinical evidence to demonstrate reasonable assurance of safety and effectiveness before market authorization. However, the true challenge extends beyond initial approval into the post-market phase. Besides, real-world device performance frequently diverges from controlled trial outcomes, exposing critical gaps in the regulatory framework, which is negatively impacting the peripheral vascular devices market growth globally.
- Economic barriers across public healthcare systems: The high cost of peripheral vascular procedures, particularly those utilizing premium-priced technologies such as drug-eluting stents (DES), drug-coated balloons (DCB), and atherectomy devices, represents a fundamental demand-side constraint in the peripheral vascular devices market. These advanced devices command significant price premiums over conventional alternatives, creating adoption barriers in cost-sensitive healthcare environments. In the Asia Pacific region, publicly funded healthcare systems and limited reimbursement frameworks severely restrict access to premium devices, thus causing a hindrance in the market expansion.
Peripheral Vascular Devices Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
6.2% |
|
Base Year Market Size (2025) |
USD 10.1 billion |
|
Forecast Year Market Size (2035) |
USD 17.3 billion |
|
Regional Scope |
|
Peripheral Vascular Devices Market Segmentation:
End user Segment Analysis
The hospitals segment in the peripheral vascular devices market is anticipated to garner the largest share of 85.2% by the end of 2035. The segment’s upliftment is highly attributed to encompassing catheterization laboratories, hybrid operating rooms, and specialized vascular surgery units within acute care hospital settings. The sustained dominance of hospitals is driven by their unique capacity to perform complex peripheral vascular interventions such as angioplasty, atherectomy, and stent placement that require advanced imaging infrastructure, including intravascular ultrasound and optical coherence tomography, along with hybrid surgical capabilities, and multidisciplinary teams comprising interventional cardiologists, vascular surgeons, and interventional radiologists.
Artery Type Segment Analysis
The iliac artery sub-segment, which is part of the artery type segment, is projected to hold the second-largest share in the peripheral vascular devices market during the forecast period. The sub-segment’s growth is highly driven by its role as a critical vessel for bifurcating from the abdominal aorta, along with acting as the ultimate blood supply conduit for lower limbs, perineum, and pelvic organs. According to official statistics published by NIH in January 2023, the internal iliac artery comprises 1 of 2 major arteries that usually arise from the common iliac artery. Besides, there exists 6 branches of the anterior division of the internal iliac artery. Moreover, the obturator artery is most frequently an early branch of the anterior division of the sub-segment, and despite this, it continues to arise from the external iliac artery through the inferior epigastric artery in an estimated 19% of individuals, thus denoting a huge growth opportunity.
Material Segment Analysis
By the end of the stipulated timeline, the nitinol segment, as part of the material, is expected to account for the third-largest share in the peripheral vascular devices market. The segment’s development is highly propelled by its unique super-elasticity and shape memory effect, permitting it to return to a pre-set shape, particularly after deformation. As per an article published by NLM in September 2022, a clinical study was conducted on 109 limbs in 103 patients to evaluate nitinol. This resulted in an overall 48 Trans-Atlantic Inter-Society Consensus (TASC) II C and 61 TASC II D lesions, along with a mean lesion length of 264 mm. Moreover, across 53% of limbs, the treatment indication catered to chronic limb-threatening ischemia, with a median follow-up of 45 months. In addition, the technical success rate was achieved for 88% of cases, thereby making it suitable for bolstering the segment’s growth.
Our in-depth analysis of the peripheral vascular devices market includes the following segments:
|
Segment |
Subsegments |
|
End user |
|
|
Artery Type |
|
|
Material |
|
|
Application |
|
|
Technology |
|
|
Product |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Peripheral Vascular Devices Market - Regional Analysis
North America Market Insights
North America in the peripheral vascular devices market is anticipated to garner the largest share of 30.5% by the end of 2035. The market’s upliftment is primarily attributed to the increased prevalence of peripheral artery disease, which is readily supported by aging demographics, along with a rise in obesity and diabetes. According to official statistics published by the CDC Government in September 2024, the obesity prevalence in the U.S. among the adult population was 40.3% in August 2023. In addition, the prevalence was higher among adults aged 40 to 59 years than among those aged 20 to 39 years and those aged more than 60 years. Besides, the disease accounted for a 9.4% prevalence, with increasingly higher rates for women than men in each of the age groups. Therefore, with such an increase in the disease, there is a huge peripheral vascular devices market demand in the overall region.
Obesity Prevalence in the U.S. Among Different Adult Age Groups (2023)
|
Age Group |
Overall Population |
Men |
Women |
|
More than 20 years |
40.3% |
39.2% |
41.3% |
|
20 to 39 years |
35.5% |
34.3% |
36.8% |
|
40 to 59 years |
46.4% |
45.4% |
47.4% |
|
More than 60 years |
38.9% |
38.0% |
39.5% |
Source: CDC Government
The peripheral vascular devices market in the U.S. is growing significantly, owing to the increased disease burden of hypertension and its associated risk factors, a shift to minimally invasive procedures, suitable reimbursement policies, innovative healthcare infrastructure, technological advancement, and the presence of notable manufacturers. As per an article published by the CDC Government in January 2025, high blood pressure has been a primary cause of 664,470 deaths in the country. In addition, 1 in 5 deaths are deliberately related to the pandemic, wherein people had a history of hypertensive disorder. Besides, almost half of adults have high blood pressure, accounting for 48.1%, which is 119.9 million. This is significantly regarded as a systolic blood pressure, which is greater than 130 mm Hg, or a diastolic blood pressure, which is more than 80 mm Hg, thus enhancing the market expansion in the overall country.
The increase in aging demographics, the increase in rare disease prevalence, generous provincial healthcare investment, the integration of minimally invasive techniques, the presence of public health system support, and growing awareness programs are certain factors that are boosting the peripheral vascular devices market in Canada. As stated in an article published by the Open Access Government in October 2025, more than 3 million people across the country are affected by over 7,000 known rare diseases, the majority of which are life-limiting, progressive, and critical. Besides, patients with atypical or rare conditions experience long-lasting diagnostic delays, usually averaging 3.7 years. However, to combat this, the country comprises a patchwork of 13 publicly funded territorial and provincial health systems, with suitable capacity, priorities, and policies that uplift the market growth.
APAC Market Insights
The Asia Pacific in the peripheral vascular devices market is expected to emerge as the fastest-growing region during the forecast period. The market’s development in the region is highly propelled by a rise in the prevalence of lifestyle-based conditions, such as high blood pressure, an increase in the aging population, a fundamental transition towards minimally invasive procedures, and technological advancements in drug-specific stents and balloons. According to official statistics published by OECD in February 2025, countries in the region have an average of 0.1 people more than 65 years for every person of working age. Besides, the increase in the elderly population originates from an increase in life expectancy, owing to optimized health and a continuous decline in birth rates. Moreover, by the end of 2060, it has been estimated that nearly 20% of the regional population in the region is poised to be aged 65 years and older, thereby denoting a huge growth opportunity for the market.
The peripheral vascular devices market in China is gaining increased traction, owing to an acceleration in approval pathways for advanced devices, an escalation in the burden of peripheral artery disease, a surge in the diabetic population, the presence of suitable government healthcare reforms, and expansion in hospital accessibility. As per an article published by NLM in June 2025, the yearly number of peripheral artery disease cases in the country significantly accounts for 2.4 million, of which 1.7 million are female, and 0.7 million are male. Besides, the number of cases for this particular disease is projected to increase to 4.0 million by the end of 2046. Therefore, to overcome this, the development of the 300 Pan-Vascular Management Center in the country has readily signaled a transition to an integrated vascular care, including management and detection of the disease, thereby making it suitable for enhancing the market demand.
The aspects of governmental expenditure on cardiovascular interventions, support by health insurance schemes for offering suitable coverage for vascular procedures, an increase in the number of patients receiving endovascular treatments, and disease awareness are a few trends that are bolstering the peripheral vascular devices market in India. As per an article published by NLM in April 2023, the healthcare system in the country is financed by different sources, with out-of-pocket spending majorly contributing 54.7% of the overall health expenditure. Besides, there exists a huge economic division in the country, with nearly 10.0% of people continuing to reside under the international poverty line of USD 2.1 per day. Therefore, with all these disparities, there is a huge growth opportunity for the market in the country.
Europe Market Insights
Europe in the peripheral vascular devices market is projected to witness considerable growth by the end of the stipulated timeline. The market’s growth in the region is highly fueled by an increase in the incidence of cardiovascular diseases, robust healthcare infrastructure across major economies, the aging demographic, well-established reimbursement policies, and an aggressive emphasis on minimally invasive procedures. According to official statistics published by OECD in December 2025, cardiovascular disease is one of the leading causes of death in the region, readily accounting for 1 in 3 of overall deaths, which is equivalent to 1.7 million deaths as of 2022, and also affecting 62 million people. Besides, ischemic heart disorder is the highest contributor to the disease, which is responsible for nearly 1/3rd, that is 547,000 deaths of total deaths, thereby making it suitable for bolstering the market growth in the overall region.
Cardiovascular Disease Mortality Analysis in Europe (2012-2022)
|
Countries |
2012 |
2022 |
|
Bulgaria |
1,074 |
1,168 |
|
Romania |
925 |
1,039 |
|
Latvia |
806 |
921 |
|
Lithuania |
784 |
901 |
|
Hungary |
732 |
809 |
|
Slovakia |
628 |
712 |
|
Croatia |
592 |
691 |
|
Estonia |
575 |
745 |
Source: OECD
The peripheral vascular devices market in Germany is gaining increased exposure, owing to the aging population, sustained need for vascular interventions, healthcare system allocation for substantial resources to interventional cardiology and vascular surgery, and the existence of rigorous safety standards, while ensuring timely market accessibility for advanced products. Based on government estimates published by the ITA in August 2025, the country has an outstanding healthcare sector, with generous expenditure per capita and employing an estimated 6.1 million people. Besides, the domestic medical device industry is considered one of the largest sectors in the country, catering to approximately USD 44 billion in yearly revenue, effectively making up 26.5% of the overall regional market. Besides, 1 in 6 employment opportunities in the country is associated with the healthcare industry, effectively generating a yearly economic footprint of USD 838 billion or an estimated 12.8% of domestic GDP, thus driving the market exposure.
The combination of universal healthcare coverage, robust governmental emphasis on advancement, and centralized procurement efficiencies, continuous patient accessibility across both rural and urban settings, are certain trends that are proliferating the peripheral vascular devices market in France. As per an article published by NLM in May 2025, there has been an increase in specialists, particularly in the country’s private sector, which operates under Sector 2, permitting them to charge fees above standard rates of 38.0% and 57.4% as of 2022. Moreover, the overall healthcare structure comprises a social health insurance that provides nearly 100% coverage for residents. Meanwhile, in terms of expenditure and financing, employee and employer payroll contributions account for approximately 33%, which is followed by 24% for general social contributions, and an estimated 20% for other taxes from value-added tax. Therefore, with the presence of such a health system, the market is gradually expanding in the country.
Key Peripheral Vascular Devices Market Players:
- Medtronic (Ireland)
- Boston Scientific Corporation (U.S.)
- Abbott Laboratories (U.S.)
- B. Braun SE (Germany)
- W. L. Gore & Associates, Inc. (U.S.)
- Edwards Lifesciences Corporation (U.S.)
- Terumo Corporation (Japan)
- Stryker Corporation (U.S.)
- BD (Becton, Dickinson and Company) (U.S.)
- Cook Group (U.S.)
- AngioDynamics, Inc. (U.S.)
- Cardiovascular Systems, Inc. (U.S.)
- LeMaitre Vascular, Inc. (U.S.)
- BIOTRONIK (Germany)
- JOTEC GmbH (Germany)
- Braile Biomédica (Brazil)
- Control Medical Technology, LLC (U.S.)
- Walk Vascular, LLC (U.S.)
- Transit Scientific (U.S.)
- Conic Vascular (U.S.)
- Siemens Healthineers (Germany)
- Mayo Clinic (U.S.)
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- Medtronic maintains a comprehensive peripheral vascular portfolio anchored by its drug-eluting stent and balloon technologies, leveraging its global scale to drive adoption across complex lesion treatments. The company emphasizes clinical evidence generation through large-scale trials to differentiate its offerings in a competitive reimbursement environment.
- Boston Scientific Corporation focuses on innovation in atherectomy and drug-eluting technologies, positioning itself as a leader in treating calcified arterial disease. The company pursues strategic acquisitions to expand its peripheral vascular capabilities and ambulatory surgery center footprint.
- Abbott Laboratories distinguishes itself through its leadership in bioresorbable scaffold technology and advanced imaging integration for peripheral interventions. The company's commitment to clinical research supports its efforts to address unmet needs in below-the-knee lesions and chronic limb-threatening ischemia.
- B. Braun SE leverages its strong presence in European healthcare systems to offer a broad range of vascular access and intervention devices with an emphasis on cost-effective solutions. The company's integrated approach combines devices with pharmaceutical offerings to support comprehensive procedural pathways.
- W. L. Gore & Associates, Inc. specializes in high-performance vascular grafts and stent grafts, leveraging its proprietary ePTFE material science for complex aneurysm and occlusive disease applications. The company maintains a focused portfolio strategy, prioritizing product durability and long-term clinical performance in niche vascular segments.
Here is a list of key players operating in the global peripheral vascular devices market:
The peripheral vascular devices market is highly competitive, characterized by a mix of global leaders and specialized innovators. Leading companies such as Medtronic, Boston Scientific, and Abbott Laboratories dominate the market with broad product portfolios and aggressive R&D investments. Key strategic initiatives include geographic expansion and acquisitions, such as Stryker’s acquisition of Inari Medical to strengthen its thrombectomy portfolio. Simultaneously, firms are focusing on next-generation technologies, such as drug-eluting stents and bioresorbable scaffolds, to improve long-term patient outcomes and secure a competitive advantage. Besides, in March 2024, BD initiated an international evaluation for expanding treatment solutions for patients with peripheral arterial disease. This evaluation is regarded as AGILITY, which assessed the effectiveness and safety of the BD vascular covered stent, thereby proliferating the peripheral vascular devices industry worldwide.
Corporate Landscape of the Peripheral Vascular Devices Market:
Recent Developments
- In February 2026, Siemens Healthineers and Mayo Clinic expanded their tactical collaboration to boost patient care for neurodegenerative disease, along with the management of metastatic liver tumors and prostate cancer by extending accessibility to the latest interventional and imaging technologies.
- In July 2024, Edwards Lifesciences made investments to effectively reflect its in-depth commitment towards advancing patient care through structural heart advancements and address large-scale unmet patient demands, as well as support sustainability long-lasting growth.
- In April 2024, Abbott received the U.S. FDA approval for its Esprit BTK Everolimus Eluting Resorbable Scaffold System, which is an outstanding innovation for patients with chronic limb-threatening ischemia below-the-knee (BTK).
- Report ID: 8487
- Published Date: Mar 30, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Peripheral Vascular Devices Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




