Medical Device Outsourcing Market Outlook:
Medical Device Outsourcing Market size was valued at USD 157.9 billion in 2025 and is projected to reach USD 493.5 billion by the end of 2035, growing steadily at a CAGR of 13.5% during the forecast period, i.e., 2026-2035. In 2026, the industry size of medical device outsourcing is assessed at USD 179.2 billion.

The rising need for operational efficiency, reduced development costs, and faster time-to-market needs among original equipment manufacturers is elevating the growth potential of the medical device outsourcing market. As medical devices become more sophisticated, manufacturers begin to depend on CDMOs and design partners to navigate complex regulatory landscapes, manage stringent quality standards, and access advanced production technologies. For example, the U.S. Food & Drug Administration (FDA) in January 2026 stated that exporting medical devices from the U.S. requires FDA-issued export certificates, now provided electronically through CECATS as downloadable PDFs since January, 2024. The FDA’s CDRH issues four types of certificates, i.e., certificates to foreign governments, certificates of exportability, and non-clinical research use only, alongside export permits and notifications. Therefore, such a regulatory environment creates an unprecedented need for medical device outsourcing.
Furthermore, the rising geriatric population, coupled with the rising occurrence of chronic diseases, is effectively fueling the demand for cardiology and orthopedic devices, thus creating an encouraging opportunity for pioneers in the medical device outsourcing market. In March 2026, the article published by the Press Information Bureau (PIB) reported that in India, there is a constant import-dependent structure that directly supports outsourcing-led manufacturing and assembly models. The report underscores that in the financial year 2022-23, exports were valued at USD 5.3 billion, whereas imports were somewhat higher at USD 17.1 billion. Also, this trend continued in FY 2023-24, wherein the exports were at USD 4.9 billion and imports at USD 16.2 billion, followed by FY 2024-25 trade flows of USD 5.1 billion in exports and USD 16.5 billion in imports. Across the entire time period, imports crossed the export values by around 3:1, thus indicating strong reliance on global sourcing of components, subassemblies, and finished devices.
India Medical Device Trade Balance with Major Global Partners 2022-2025: Export-Import Trends in USD Billion
|
Country |
2022-23 |
2023-24 |
2024-25 |
|
U.S. Export |
0.75 |
0.77 |
0.80 |
|
U.S. Import |
1.62 |
1.55 |
1.45 |
|
Germany Export |
0.29 |
0.28 |
0.30 |
|
Germany Import |
0.96 |
0.91 |
0.94 |
|
France Export |
0.17 |
0.16 |
0.17 |
|
France Import |
0.24 |
0.23 |
0.23 |
|
China Export |
0.17 |
0.16 |
0.16 |
|
China Import |
1.25 |
1.18 |
1.19 |
|
Netherlands Export |
0.15 |
0.15 |
0.15 |
|
Netherlands Import |
0.42 |
0.40 |
0.44 |
Source: PIB
Key Medical Device Outsourcing Market Insights Summary:
Regional Highlights:
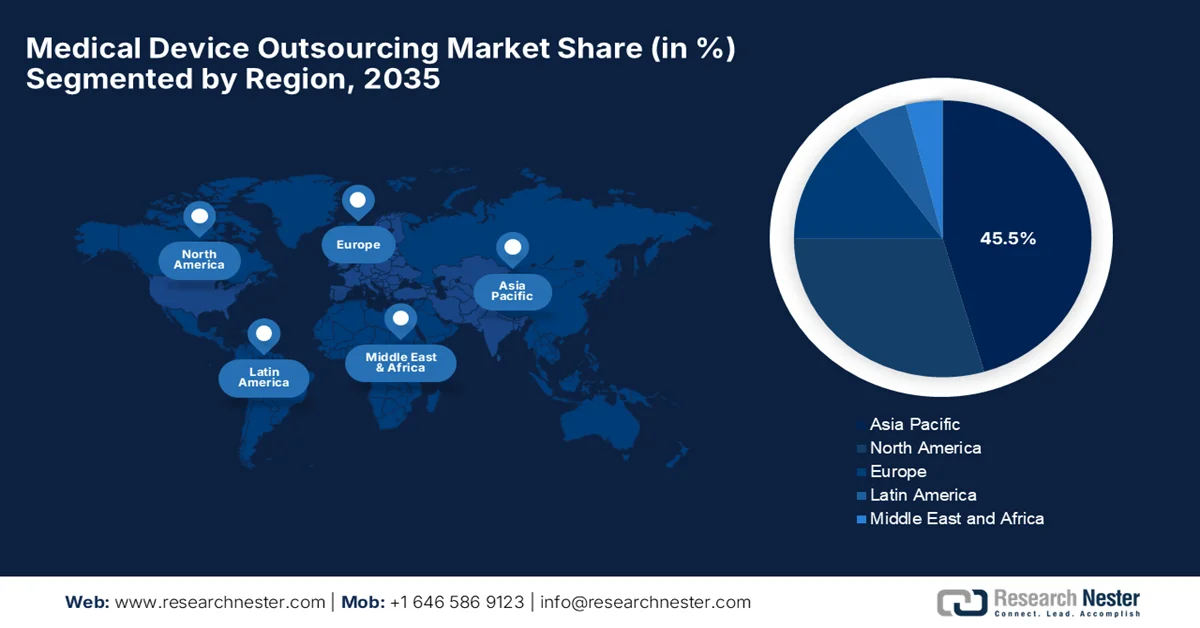
- Asia Pacific is projected to hold a dominant 45.5% share by 2035 in the medical device outsourcing market, fueled by cost advantages, expanding manufacturing capabilities, and rising demand for affordable medical devices
- North America is expected to witness notable growth through 2026-2035, attributed to increasing reliance on specialized outsourcing partners amid rising R&D costs and strong presence of medical device companies
Segment Insights:
- In the medical device outsourcing market, contract manufacturing under the service segment is anticipated to capture a leading 55.4% share by 2035, propelled by rising demand for scalable, cost-efficient production alongside stringent quality system compliance requirements
- Class II under the device class segment is projected to gain a considerable share during 2026-2035, driven by moderate regulatory controls and widespread outsourcing suitability of standardized medical devices
Key Growth Trends:
- Rising cost pressure on manufacturers
- Technological advancements in manufacturing
Major Challenges:
- Regulatory complexity and compliance burden
- Intellectual property and data security concerns
Key Players: IQVIA Holdings Inc. (U.S.), Charles River Laboratories International, Inc. (U.S.), Thermo Fisher Scientific Inc. (U.S.), Integer Holdings Corporation (U.S.), Jabil Inc. (U.S.), Plexus Corp. (U.S.), STERIS plc (Ireland), Parexel International Corporation (U.S.), Eurofins Scientific SE (Luxembourg), SGS SA (Switzerland), Intertek Group plc (UK), Lonza Group AG (Switzerland), ELOS Medtech AB (Sweden), AcuityMD (U.S.), Sanner GmbH (Germany).
Global Medical Device Outsourcing Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 157.9 billion
- 2026 Market Size: USD 179.2 billion
- Projected Market Size: USD 493.5 billion by 2035
- Growth Forecasts: 13.5% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: Asia Pacific (45.5% Share by 2035)
- Fastest Growing Region: North America
- Dominating Countries: United States, China, Germany, Japan, India
- Emerging Countries: South Korea, Brazil, Mexico, Singapore, Ireland
Last updated on : 28 April, 2026
Medical Device Outsourcing Market - Growth Drivers and Challenges
Growth Drivers
- Rising cost pressure on manufacturers: The medical device companies are opting for outsourcing to reduce operational and production costs. The high expenses which are related to R&D, manufacturing infrastructure, and labor encourage firms toward third-party providers. In March 2026, Eurofins announced the launch of Eurofins Medical Device Consulting in the U.S., making an expansion from testing services to full-lifecycle MedTech strategy and policy support. The new chapter focuses on regulatory intelligence, product development, and government relations, helping clients navigate through the U.S. FDA authorization and beyond. This investment positions Eurofins as a partner in commercial strategy, compliance, and innovation, turning quality data into understanding across the entire product lifecycle, thus positively impacting the medical device outsourcing market’s growth and exposure.
- Technological advancements in manufacturing: The improvements in terms of technologies such as automation, AI, and precision engineering are catalyzing demand in the medical device outsourcing market. Specialized contract manufacturers are making strong investments in advanced technologies, allowing medical device companies to access efficient production capabilities. In June 2025, Phillips Medisize announced the launch of Theravolt medical connectors, which are especially engineered to enhance device integration, reliability, and performance in next-gen medical technologies. The connectors are designed for high pin count and voltage clearance, and it supports advanced applications such as electrophysiology and pulsed field ablation, simplifying integration while reducing costs and customization time. Hence, such instances will drive medical device outsourcing market growth by enabling more reliable medical device integration and accelerating innovation in healthcare technologies.
- Growing demand for medical devices: The rising healthcare awareness is boosting demand for medical devices globally. This surge compels manufacturers to scale production quickly, which is efficiently achieved through outsourcing partners with established infrastructure as well as flexible manufacturing capabilities. As per an article published by the International Trade Administration (ITA) in March 2025, India’s medical device sector is anticipated to rise from USD 12 billion in 2023-24 to a substantial USD 50 billion by the end of 2030, owing to the rising healthcare demand, chronic diseases, and government initiatives. It also mentions that the U.S. MedTech firms have strong export potential, with U.S. exports to India rising 66.3% in FY24 to USD 1.45 billion. In this context, success will depend on strategic partnerships, regulatory compliance, and delivering cost-effective, high-quality innovations, thus benefiting the medical device outsourcing market.
Historic Global Medical Devices Market Share and CAGR by Region - Industry Breakdown 2021
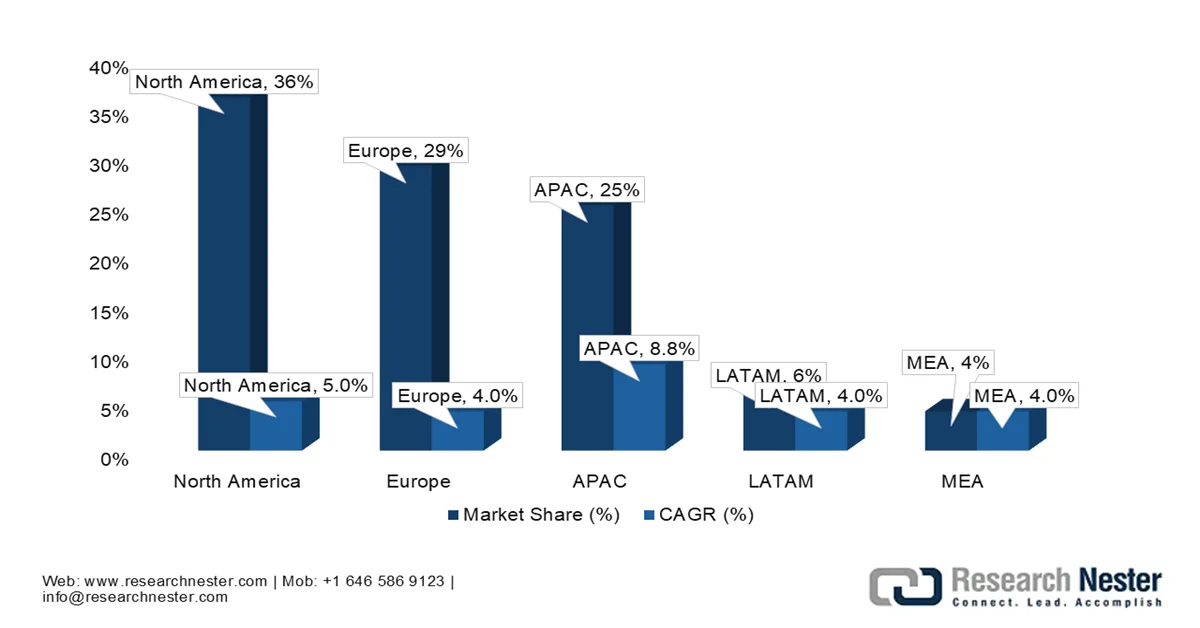
Source: APACMed.org
Challenges
- Regulatory complexity and compliance burden: Medical device outsourcing market growth is being hindered due to the complexity of regulatory bodies across different nations. In this context, outsourcing partners need to adapt to continuously evolving standards for safety, quality, and documentation, which increases operational costs and delays timelines. Apart from this, any minor compliance failures can result in product recalls, penalties, or restrictions in terms of market access. In addition, differing regulations across regions create fragmentation, thereby forcing outsourcing providers to maintain multiple compliance systems. This makes scalability difficult and requires continuous investment in regulatory expertise, training, and infrastructure, especially for companies that are serving global clients.
- Intellectual property and data security concerns: Outsourcing some of the critical stages, such as design, prototyping, and manufacturing, raises concerns around intellectual property protection and data security. Medical device companies operating in the medical device outsourcing market usually share proprietary technologies along with sensitive clinical data with third-party vendors, which increases the risk of IP leakage or cyberattacks. This is especially challenging when outsourcing to regions with changing levels of IP enforcement and cybersecurity standards. At the same time, the rise of digital health and connected devices amplifies the risks associated with patient data protection and compliance with data privacy regulations. Therefore, making sure regarding secure data exchange, robust contractual agreements, and strict vendor audits becomes highly essential, but also adds complexity and cost to outsourcing relationships.
Medical Device Outsourcing Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
13.5% |
|
Base Year Market Size (2025) |
USD 157.9 billion |
|
Forecast Year Market Size (2035) |
USD 493.5 billion |
|
Regional Scope |
|
Medical Device Outsourcing Market Segmentation:
Service Segment Analysis
Contract manufacturing in the service segment is expected to dominate the medical device outsourcing market with the largest revenue share of 55.4% during the forecast period. The dominance of the segment is largely propelled by rising demand for scalable, cost-efficient production and strict compliance requirements enforced under quality system regulations. High complexity in device design and the need for precision engineering also solidify dependence on external manufacturers with advanced capabilities and global supply networks. In May 2025, Quasar Medical signed an agreement to acquire Nordson MEDICAL’s design and development businesses in Galway, Ireland, and Tecate, Mexico, with the main goal to strengthen its position as a global full-lifecycle CDMO. This acquisition adds FDA-registered and ISO-certified facilities with strong knowledge in balloon and catheter design, prototyping, and scalable assembly, thereby expanding Quasar’s footprint to 10 facilities worldwide.
Device Class Segment Analysis
Under the device class segment, class II is anticipated to garner a considerable share in the medical device outsourcing market during the stipulated timeframe. They include widely used products such as infusion pumps, diagnostic equipment, and surgical instruments. These devices necessitate moderate regulatory controls, making them suitable for outsourcing manufacturing and testing. In January 2022, the Prometra programmable infusion pump system, which is a Class II medical device developed by Flowonix Medical, Inc. and approved by the U.S. FDA for intrathecal drug delivery, includes baclofen for patients who are aged 12 and above. As a programmable infusion pump, it exemplifies a widely used Class II device, making it well-suited for outsourced manufacturing and validation activities. Therefore, this reflects the visible industry trend wherein Class II devices, such as infusion pumps, are most frequently outsourced due to their standardized regulatory pathways and repeatable production requirements.
Application Segment Analysis
By the end of 2035, the cardiology device segment is forecasted for a noteworthy expansion in the medical device outsourcing market. The rising global occurrence of cardiovascular diseases is driving continuous innovation in stents, pacemakers, and monitoring devices. This high demand encourages outsourcing of design, manufacturing, and testing to specialized providers who are capable of meeting both strict safety and performance standards. In May 2023, the U.S. FDA approved Abbott’s TactiFlex ablation catheter, sensor-enabled, the world’s first ever catheter with a flexible electrode tip and contact force sensing for treating atrial fibrillation. It is used with Abbott’s EnSite X EP system, and it provides enhanced visualization, accuracy, and stability, reducing procedure times and improving safety. Apart from this its development and clinical validation across international studies show that modern cardiology devices rely on distributed manufacturing and testing to meet required standards.
Our in-depth analysis of the medical device outsourcing market includes the following segments:
|
Segment |
Subsegments |
|
Service |
|
|
Device Class |
|
|
Application |
|
|
Outsourcing Model |
|
|
End user |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Medical Device Outsourcing Market - Regional Analysis
APAC Market Insights
The Asia Pacific medical device outsourcing market is anticipated to secure the largest share of 45.5% by the conclusion of the forecast period. The region’s growth in this field is effectively fueled by cost advantages, expanding manufacturing capabilities, and increasing demand for affordable medical devices. Countries such as China and India are major outsourcing hubs, whereas Japan and South Korea contribute through advanced technology and precision manufacturing. Based on the government data published in April 2022, the region accounts for approximately 28% of the global medical devices industry, which makes it one of the largest regional contributors. It mentions that the Asia Pacific is growing at a CAGR of about 6% to 7%, owing to the expanding healthcare infrastructure and demand for affordable devices, thus denoting a positive medical device outsourcing market outlook.
Japan Medical Device Industry: Production, Imports, Exports & Trade Trends 2022 to 2024 (USD Million)
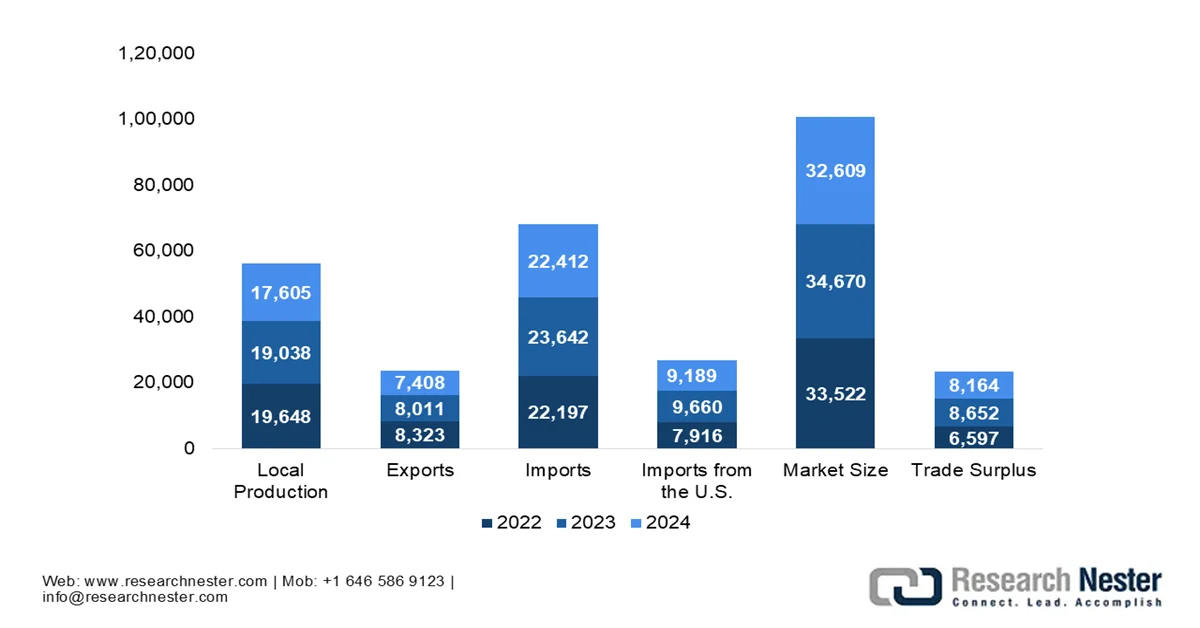
Source: ITA
The strong manufacturing base, growing demand for advanced and effective therapies, and increased investment in R&D for innovative treatments are certain factors driving growth in the China medical device outsourcing market. In addition to supportive government policies, a large patient pool, and the availability of cost-efficient contract manufacturers, further solidify its leading position in the regional market. Based on the government data published in January 2026, the country has expanded its bulk procurement program for surgical devices, with the main goal to reduce healthcare costs for hospitals and patients nationwide. The sixth round included 440 products from 202 manufacturers, covering cardiovascular tools such as drug-coated balloons and, for the first time, devices for urinary stone surgeries such as guidewires and balloon catheters. This readily broadens the centralized procurement to 142 types of consumables across nine categories, and authorities expect significant cost reductions.
Enhanced by cost-effective production capabilities and skilled technical expertise, the medical device outsourcing market in India is expected to witness solid growth in the next decade. Key players in this field are outsourcing contract manufacturing, product design, and regulatory compliance services to optimize operational costs and accelerate development. The growth is supported by government initiatives encouraging domestic production. As per the article published by PIB in December 2025, India’s national medical devices policy, 2023, is efficiently driving rapid growth in the sector through strategies in regulation, infrastructure, R&D, investment, workforce development, and global branding. It also mentions the key initiatives of the PLI scheme with a USD 410 million outlay, commissioning 22 greenfield projects and boosting domestic production of high-end devices, along with the creation of medical device parks in Noida, Ujjain, and Kanchipuram. These measures have raised exports from USD 2.9 billion in FY22 to USD 4.1 billion in FY25, thus denoting a positive medical device outsourcing market outlook.
North America Market Insights
The North America medical device outsourcing market is expected to witness considerable growth over the forecast period. The region’s prominence in this field is mainly attributable to the strong presence of medical device companies that increasingly outsource regulatory and consulting activities to specialized service providers. In addition, the burgeoning cost of R&D has prompted many companies in the region to rely on third-party vendors, further supporting market growth. In March 2025, Flex announced the product introduction center near Boston to support healthcare customers with end-to-end product development, from prototype to preclinical builds, design verification, and production transfer. This particular facility consists of an 8,000 sq. ft. NPI lab and a 2,000 sq. ft. quality testing lab, offering design-for-excellence services to ensure manufacturing readiness.
The need for efficient raw material management systems and high maintenance requirements is responsibly driving the upliftment of the U.S. medical device outsourcing market. The country’s market also benefits from the need for regulatory compliance, cost optimization, and access to specialized manufacturing technology. For instance, in November 2023, Millstone Medical Outsourcing announced the relocation of its testing services division to a newly expanded 25,000 sq. ft. facility in Bloomfield, Connecticut, representing a 400% increase in capacity. This move follows its 2022 acquisition of Ethide Laboratories and MycoScience, bringing decades of expertise in microbiology and environmental testing. Hence, these constant efforts from the leading pioneers will position the country as a predominant leader in the global dynamics of the medical device outsourcing industry.
The rising need for specialized manufacturing processes, coupled with a strong regulatory proficiency among domestic small-and medium-sized enterprises and global partners, is responsibly fueling the medical device outsourcing market in Canada. In addition, the need for accelerated market debuts and rising demand for advanced technologies, including wearable medical devices and IoT-enabled systems, are fueling the market’s growth. In December 2023, the country’s government amended its medical devices regulations with the main goal of broadening provisions for devices needed during urgent public health situations. These changes remove COVID-19-specific definitions and establish a proper framework for a wider range of conditions, thereby allowing the Ministry of Health to add medical devices or categories to an official list when immediate action is required. Hence, these updates strengthen administrative understanding, ensuring rapid access to critical devices in emergencies.
Europe Market Insights
Europe medical device outsourcing market is expected to witness considerable growth during the stipulated timeframe. The region’s growth in this field is largely driven by the strong regulatory framework and presence of leading testing and certification providers. Growth is also driven by increasing outsourcing related to compliance with stringent regulations, such as EU MDR, and the expansion of quality assurance services. In January 2025, Arterex announced the acquisition of Phoenix S.r.l., which is an Italy-based medical device developer and manufacturer, expanding its capabilities in Europe. Phoenix has expertise in disposable device design, assembly, and packaging with ISO 13485 certification and EU MDR compliance, operating from a 37,000 sq. ft. facility with ISO 8 clean rooms. From a strategic perspective, such instances enhance end-to-end services for medical device customers across the region’s vast geography.
The medical device outsourcing market in Germany accounts for a surging expansion efficiently supported by a well-established device industry, strong emphasis on innovation, and advanced manufacturing capabilities. In addition, continued R&D investments in respiratory therapies and stringent regulatory requirements for quality and safety are encouraging companies to collaborate with well-experienced contract manufacturers. ITA in August 2025 revealed that the country has the third-largest medical technology market worldwide, generating about USD 44 billion annually, accounting for 26.5% of the regional market. It also mentioned that healthcare export value reached USD 172 billion in 2023, whereas imports were valued at USD 188.5 billion, reflecting strong international trade activity. Key segments such as single-use medical devices are rapidly expanding, projected to grow from USD 18.6 billion in 2023 to USD 53.4 billion by 2030 with a CAGR 16.3%, thus denoting a positive medical device outsourcing market outlook.
Germany Medical Equipment Market Size, Imports, Exports & Trade Balance 2022-2024: Historical Data & Forecast (USD Billion)
|
Category |
2022 |
2023 |
2024 |
|
Total Exports |
27.1 |
30.0 |
31.0 |
|
Total Imports |
24.0 |
25.3 |
26.0 |
|
Imports from the U.S. |
5.2 |
5.6 |
6.0 |
|
Trade Surplus |
-2.24 |
-0.73 |
-6.0 |
|
Exchange Rate |
1.053 |
1.0813 |
1.0824 |
Source: ITA
The UK medical device outsourcing market is poised for solid growth in the next decade, owing to the need for faster product development, regulatory compliance, and adoption of advanced technologies such as automation and AI in manufacturing. Outsourcing partners are utilized for contract manufacturing, design, and navigating complex post-Brexit regulatory requirements. In February 2026, the country’s government data stated that MHRA approved a record number of medical device clinical investigations in 2025, rising 17% when compared to the previous year and giving patients earlier access to breakthrough technologies. Growth was largely driven by neurotechnology, wherein the studies doubled since 2024, along with rapid expansion in AI-powered devices for imaging, treatment guidance, and personalized care. The MHRA met all statutory deadlines, thereby reducing approval times to an average of 51 days, which is suitable for bolstering the overall country’s market growth.
Key Medical Device Outsourcing Market Players:
- IQVIA Holdings Inc. (U.S.)
- Charles River Laboratories International, Inc. (U.S.)
- Thermo Fisher Scientific Inc. (U.S.)
- Integer Holdings Corporation (U.S.)
- Jabil Inc. (U.S.)
- Plexus Corp. (U.S.)
- STERIS plc (Ireland)
- Parexel International Corporation (U.S.)
- Eurofins Scientific SE (Luxembourg)
- SGS SA (Switzerland)
- Intertek Group plc (UK)
- Lonza Group AG (Switzerland)
- ELOS Medtech AB (Sweden)
- AcuityMD (U.S.)
- Sanner GmbH (Germany)
- WuXi AppTec Co., Ltd. (China)
- Samsung Biologics Co., Ltd. (South Korea)
- Fujifilm Holdings Corporation (Japan)
- Avania (Netherlands)
- Jubilant Biosys Limited (India)
- Nova Laboratories Sdn Bhd (Malaysia)
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- IQVIA Holdings Inc. is identified as a dominant player in this field, especially in terms of clinical research, regulatory support, and real-world evidence services for medical devices. The company differentiates itself by incorporating advanced analytics, AI-based platforms, and strong clinical trial infrastructure spanning across the globe.
- Thermo Fisher Scientific Inc. is leading the medical device outsourcing market, propelled by its extensive CDMO capabilities, which support both biopharma and medical device companies. The firm deliberately leverages its vast global manufacturing network, strong supply chain infrastructure, and integrated service portfolio, which spans research, development, and production.
- WuXi AppTec Co., Ltd. has emerged as a major global outsourcing partner by building a fully integrated CRDMO platform that spans research, development, testing, and manufacturing. Its rapid global expansion, especially in the U.S., China, and Europe, efficiently supports scalable outsourcing solutions.
- Eurofins Scientific SE is a leading provider of testing, inspection, and certification services for medical devices, which has strong emphasis on regulatory compliance. Besides, the firm has expanded aggressively through acquisitions and infrastructure investments, building one of the largest global laboratory networks.
- SGS SA is a global leader in testing, inspection, and certification services, which benefits from a strong foothold in medical device regulatory outsourcing. The company’s competitive advantage depends on its notified body status in Europe, which enables it to support CE marking and compliance under Europe’s MDR regulations.
Below is the list of some prominent players operating in the global medical device outsourcing market:
The competitive landscape of the medical device outsourcing market is mostly consolidated, wherein the global players are intensely competing in terms of integrated service offerings, regulatory support, and technological capabilities. At the same time, companies such as IQVIA, WuXi AppTec, and Thermo Fisher are making heavy investments in terms of end-to-end platforms by combining research, development, and manufacturing, whereas firms such as SGS and Eurofins are highly focused on expanding testing and compliance services. Apart from these strategic initiatives, the players adopted acquisitions, geographic expansion, and the integration of AI and digital tools with a collective goal to improve efficiency and compliance. In September 2025, IQVIA introduced the clinical trial financial suite, which is an AI-based platform that unifies budgeting, contracting, forecasting, and payments with the main goal of streamlining financial management in clinical trials.
Corporate Landscape of the Medical Device Outsourcing Market:
Recent Developments
- In April 2026, AcuityMD secured a total of USD 80 million in Series C funding, thereby bringing its valuation to USD 955 million and fueling the launch of AcuityAI, which is an agentic AI platform suitable for medtech. It uses a proprietary healthcare knowledge graph to help medical device companies make faster, data-driven sales and market decisions.
- In March 2026, IQVIA announced the launch of IQVIA.ai, which is a unified agentic AI platform powered by NVIDIA, designed to transform operations, decision-making, and innovation across life sciences. The platform combines IQVIA’s healthcare-grade AI and data assets with NVIDIA’s advanced AI technologies.
- In October 2025, WuXi AppTec signed strategic MoUs with NEOM and Saudi Arabia’s Ministry of Health to extend its global CRDMO platform into the region. This partnership with NEOM focuses on establishing pharmaceutical R&D and manufacturing facilities at Oxagon.
- Report ID: 8538
- Published Date: Apr 28, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Medical Device Outsourcing Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




