Medical Device Testing Market Outlook:
Medical Device Testing Market size was over USD 11.1 billion in 2025 and is estimated to reach USD 25.3 billion by the end of 2035, expanding at a CAGR of 9.6% during the forecast timeline, i.e., 2026-2035. In 2026, the industry size of medical device testing is assessed at USD 12.7 billion.

The rising regulatory requirements in countries are propelling the growth of the medical device testing market. FDA, EMA and MHRA demand certification and test results from manufacturers, leading to greater adoption of medical device testing. According to data from the World Health Organisation, countries spend more than USD 30.5 billion every year on substandard medical products, including medical devices, resulting from poor testing.
Healthcare facilities and diagnostic centres largely depend on medical devices such as MRI machines and ultrasound machines to scan various organs of the human body. Faulty medical devices can lead to inaccurate diagnosis leading to negative patient outcomes. Cardiovascular cases across the globe are rising, demanding accurate radiological images to determine the threat level. The growth of the market extends improvement in the accuracy of devices, leading to effective preventive care. Regions such as the Asia Pacific are supporting the market expansion because of low-cost servicing and skilled technicians.
Key Medical Device Testing Market Insights Summary:
Regional Highlights:
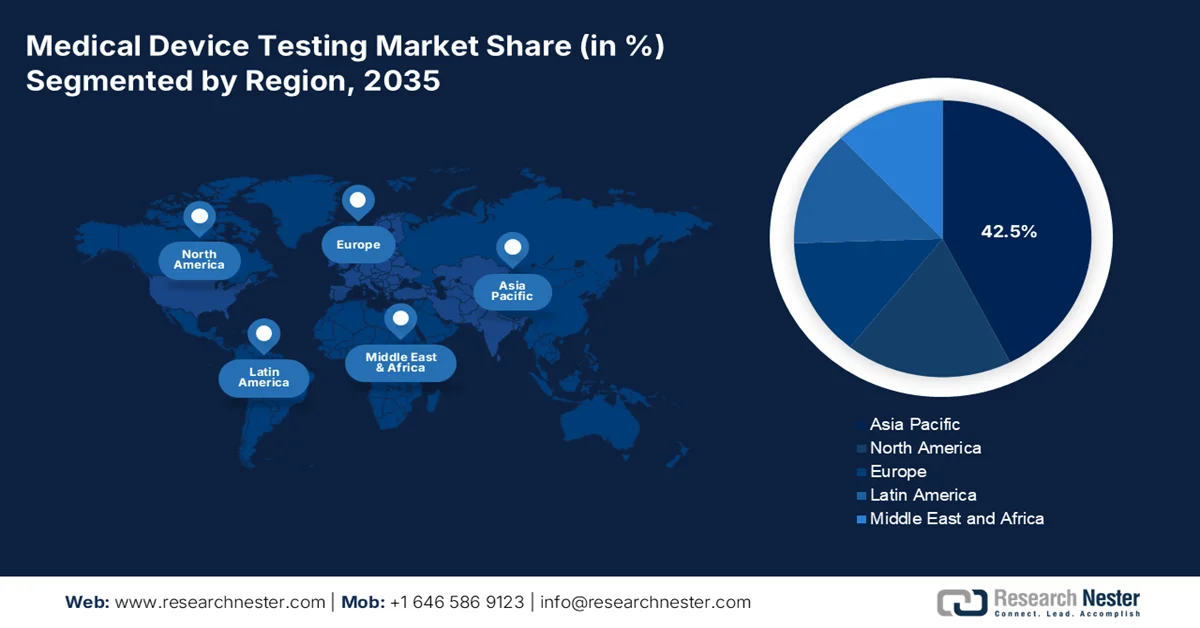
- The Asia Pacific medical device testing Market is forecast to secure a commanding 42.5% share by 2035, supported by cost-efficient manufacturing ecosystems and rising healthcare demand across a large patient population.
- North America is projected to maintain a substantial share by 2035, reinforced by strong research capabilities and regulatory-led testing requirements for advanced imaging and diagnostic devices.
Segment Insights:
- Clinical Segment: Within the medical device testing market, the clinical segment is projected to account for a dominant 60.5% share by 2035, as it represents the final and most critical approval stage requiring extensive human trials in line with stringent FDA and EMA regulatory mandates.
- Biocompatibility Test Segment: The biocompatibility test segment is expected to command the largest share by 2035, supported by its essential role in verifying device safety, toxicity-free composition, and regulatory compliance to minimize patient risk.
Key Growth Trends:
- Rise in digital health devices
- Investment in the healthcare sector
Major Challenges:
- High cost of testing
- Supply chain constraints
Key Players: TÜV SÜD (Germany), DEKRA (Germany), BSI Group (United Kingdom), Intertek (United Kingdom), UL (Underwriters Laboratories) (U.S.), Eurofins Scientific (Luxembourg), Medistri (Switzerland), NAMSA (U.S.), Eurofins BioPharma Services (U.S.), Covance (U.S.)
Global Medical Device Testing Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 11.1 billion
- 2026 Market Size: USD 12.7 billion
- Projected Market Size: USD 25.3 billion by 2035
- Growth Forecasts: 9.6% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: Asia Pacific (42.5% share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, China, Germany, Japan, United Kingdom
- Emerging Countries: India, Brazil, South Korea, Mexico, Singapore
Last updated on : 10 February, 2026
Medical Device Testing Market - Growth Drivers and Challenges
Growth Drivers
- Rise in digital health devices: The global rise in population is leading to a higher prevalence of chronic diseases, which is resulting in higher adoption of digital health devices such as smart bands that allow practitioners to monitor patients remotely. The wearables and the digital health devices undergo strong iterations and modifications to reduce faults within the products. Manufacturers often test the products in-house to limit the expenses, where hardware and software are extensively checked to maintain continuous performance. Emerging economies such as China and India are heavily manufacturing medical devices that are demanding iterations and testing, leading to higher adoption in the market. According to the National Library of Medicine, 21% of the U.S. population wears fitness trackers, which demand accurate measurements, demanding rigorous testing, supporting the expansion of the market.
- Investment in the healthcare sector: The demand for the market is driven by the large-scale investment made by the government and other agencies to promote diagnostic effectiveness and bring positive patient outcomes. According to OECD data of 2023, more than 11.5% of the total GDP is spent on improving the healthcare infrastructure, showing the demand for medical devices, resulting in increased testing and iteration. Japan’s medical infrastructure is dependent on technology, where complex, ultra-modern medical devices are used. These systems are often used in critical areas such as the resuscitation and emergency rooms, which support sustaining lives. Thus, maintaining accuracy and precision in such systems becomes essential, which amplifies the growth of the market
- Technological Modifications: Globally, medical devices are being innovated, which is giving rise to effective treatments and diagnoses of chronic diseases. Manufacturers have integrated AI and cloud computing into medical devices, propelling the growth of the market. The ultra-modern medical devices need to undergo testing to ensure the accurate integration of software and hardware. Robotic-assisted surgery equipment is being adopted across healthcare facilities to minimise surgery pain and gain supreme diagnostic efficiency, which demands extensive testing and modifications to reduce errors and maintain uniformity across surgeries, propelling the expansion of the market.
Challenges
- High cost of testing: The testing of the medical devices can be quite expensive, which leads to a decline in the growth of the market. Moreover, manufacturers prefer testing their product in-house to reduce cost, which further leads to a limited outsourcing scope, thereby declining the growth of the market. The rising compliance among the agencies is leading to increased wait time, elevating the cost of testing.
- Supply chain constraints: Testing medical devices demands the use of certain equipment, which is often challenging to procure because of geopolitical issues. The availability of the materials has severely impacted the growth of the market, leading to a decline in the adoption of medical device testing. Manufacturers often outsource the testing to reputed labs, which are inundated with surging demand, which can cause delays in testing, further slowing down the growth of the market.
Medical Device Testing Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
9.6% |
|
Base Year Market Size (2025) |
USD 11.1 billion |
|
Forecast Year Market Size (2035) |
USD 25.3 billion |
|
Regional Scope |
|
Medical Device Testing Market Segmentation:
Phase Type Segment Analysis
The clinical segment is anticipated to hold the largest share of 60.5% by the end of 2035 due to the rapid development of medical devices, where clinical trials are considered the last stage for approval. The medical devices are tested on humans to enhance assessment and their usability. The tightening global regulations from agencies such as the FDA and EMA demand extensive assessment and clinical approvals to ensure minimal impact on the patients and enhance the point of care. The clinical trials are widely considered safe, after which the products are supplied to the market, enhancing the growth of the medical device testing market. Wearable technologies are often directly exposed to the skin, which can cause shocks because of loose terminals, which demands testing to ensure the safety and efficacy of the devices.
Service Type Segment Analysis
The biocompatibility test will hold the largest share owing to its importance in testing the medical devices’ safety and accuracy. Biocompatibility tests often assess the products comprehensively, which reduces the potential risk. The biocompatibility tests ensure the medical devices are free of toxins, mutagenic and immunogenic components that will not harm the patient adversely. The regulatory body demands that the products undergo rigorous testing, including a biocompatibility test, to further eliminate the chances of infections and irritation while using the product. The biocompatibility tests undertaken by the device manufacturers are significantly increasing the scope of the market.
Our in-depth analysis of the global market includes the following segments:
|
Segment |
Subsegments |
|
Service Type |
|
|
Phase Type |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Medical Device Testing Market - Regional Analysis
Asia Pacific Market Insights
APAC market is expected to hold a 42.5% share due to the low-cost manufacturing of testing components, enabling players to drive global demand for medical device testing. In 2024, Stryker launched its facility that would cater to lifecycle testing of medical devices to reduce potential risk and understand efficacy. The new laboratory features skilled microbiologists who are capable of extensively testing the microbiology of the products, enhancing the safety of the products. The large population of the region has increased the prevalence of diseases, making the need for accurate systems essential, which in turn propels the growth of the Asia Pacific market.
China has a strong ecosystem of manufacturing because of the low cost of components and labour, propelling the electrical and electronics market. The steady growth of the electrical and electronics market is amplifying the growth of the medical device market, that demand rigorous testing and certifications. The low-cost manufacturing and steady growth of the medical device production are expanding the market. China is leading in terms of population, where the prevalence of chronic diseases is increasing, demanding accurate results upon testing, further propelling the market of medical device testing.
India is growing in terms of healthcare infrastructure, especially after the pandemic. The country is now developing hospitals and diagnostic centres to meet the rising demand. The clinics and hospitals employ a large number of medical devices, such as MRI and CT scan systems, which generate images of the human body components. These systems demand extensive testing in terms of features and their performance in emergencies. The presence of multiple key players within the country encourages the manufacturers to undergo testing and obtain a certification of quality.
North America Market Insights
North America holds strong research and innovation skills, fueling the growth of advanced medical devices that require testing to measure efficacy. The region also holds large number of residents with varied medical needs demanding imaging and scanning through MRI and CT scans, enabling effective detection of diseases. Such devices demand testing in order to provide patient-centric care and enable positive patient outcomes. Agencies and regulatory bodies pressure manufacturers to submit testing certifications, which ensure accuracy and biocompatibility, by reducing the potential risks of the device. The innovation skills and large population are significantly driving the North America market for medical device testing.
U.S. FDA has laid strong policies and regulations that standardise device testing and obtaining a clearance certificate, helpful in assuring the quality and performance. The US is witnessing a rise in disposable income, which is pushing patients to avail premium healthcare, which has propelled the adoption of medical devices. The large number of hospitals and clinics has also introduced medical devices that help meet the rising demand from the population. The performance testing of such devices will support in decision making, enabling better health for the patients. The efforts of the FDA and the rising disposable income of the country are propelling the growth of the medical testing device market in the U.S.
The medical device testing market in Canada is driven by regulatory compliance, innovation and patient safety, which enhances patient-centric care. The country focuses on conducting clinical trials for the medical devices that help in assessing their suitability and their responsiveness towards humans. The prolonged clinical trials enhance the performance of the devices and ensure minimal risk upon use. It also measures the post-market surveillance, where patient feedback and their responses are recorded to understand the effectiveness of the devices and make further iterations to provide patients with uniform care.
Europe Market Insights
Europe medical device testing market is expected to hold a significant share by the end of 2035, owing to innovation and growth in regulations. Europe is strict with medical product regulations were manufacturers need to submit proof of efficacy to improve patient-centric care. The innovation within the region is also accelerating the growth of the market. AI and cloud computing within medical devices have demanded extensive checking and iteration to eliminate any errors during the patient's use. NHS and other agencies are encouraging the remote monitoring of patients, requiring health trackers that need to maintain accuracy, based on which an effective diagnosis can be made.
The UK holds a large number of hospitals and clinics aimed at improving accessibility, leading to the growth of medical devices. These devices undergo testing from outsourced companies to assess their features and their effectiveness in patient-centric care. The rise in production is also one of the driving factors for medical device testing in the country. As manufacturers impose innovation within their portfolio, the demand for medical device testing will increase. As per the regulations of the UK, ISO10993 is essential for the product which required extensve testing of the product and assessing the biocompatibility of the product. The rising healthcare infrastructure and regulations from the government are amplifying the growth of the UK market.
Germany medical device testing market witnesses certain policies and protocols laid for digital health monitoring devices, where the products need to be tested by government agencies to determine the performance and biocompatibility. The country prefers clinical trials for every medical product used in critical and complex procedures such as cardiological and oncological surgeries. Robot-assisted surgeries are rising in Germany, which is fueling the growth of the market. The tests also include sampling within the patients to understand the accuracy and effectiveness of the medical devices.
Key Medical Device Testing Market Players:
- TÜV SÜD (Germany)
- DEKRA (Germany)
- BSI Group (United Kingdom)
- Intertek (United Kingdom)
- UL (Underwriters Laboratories) (U.S.)
- Eurofins Scientific (Luxembourg)
- Medistri (Switzerland)
- NAMSA (U.S)
- Eurofins BioPharma Services (U.S.)
- Covance (U.S.)
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
-
The players operating in the medical device testing market are expected to face intense competition during the forecast period. The market is associated with both established key players and new entrants. Nevertheless, the market is moderate. New players impose huge competition for the pre-existing players, prohibiting them from acquiring the majority of the revenue share. Specialised manufacturers maintain a competitive landscape in the market. Key players in the market are significantly supported by the governments for research and innovation.
Corporate Landscape of Medical Device Testing Market:
- TUV SUD: An organisation that specialises in testing and quality management services for medical devices. The business conducts clinical and biocompatibility trials that help assess product quality and effectiveness. The business is also compliant with various regulatory bodies such as the FDA and EMA, which enables effective trials of the medical devices.
- DEKRA: A leading provider of medical device testing services with a wide range of portfolio, including testing for regulatory compliance, biocompatibility and clinical trials. The business services several industries, including automotive, healthcare and electronics, which demand rigorous testing of their products. The risk assessment and the quality assurance services are widely used by manufacturers to eliminate potential risks of the products.
- BSI Group: The business is one of the oldest and most prominent service providers with a long legacy of standardised testing. The business offers medical device testing with special focus on regulatory approvals, biocompatibility tests and ISO certifications, which are accredited by Europe and the FDA. BSI group has a presence in multiple countries because of their core understanding in medical device approval process.
- Intertek: A quality assurance and certification business based in UK that serves various industries, including healthcare and automotive. The business focuses on product quality testing, compliance testing, and biocompatibility testing, which enables product certifications and their adoption in the market. Intertek encourages global manufacturers to test their products and obtain certification.
- Underwriters Laboratories: A global safety science company that focuses on testing, certifications and inspection of medical devices and a range of other products. The business also serves as an electrical and material testing facility for medical devices, which further enhances the safety of the products. UL is widely recognised by the FDA and EMA, which act as the key regulatory bodies to approve medical products.
Here is a list of key players operating in the global market:
Recent Developments
- In January 2025, Eurofins launched its new package testing lab to enhance the scope of the medical device testing market. The new facility will measure the biocompatibility and sterility of the products. The facility will enhance the regulatory testing across the US, Europe and the Asia Pacific.
- In January 2025, Medistri announced expansion in Hungary to improve access to healthcare solutions. The new facility provided a sterilisation capacity of more than 15,000 pallets per year, demonstrating domination over demands. The in-house laboratory services will ensure seamless quality control and operational efficiency.
- Report ID: 3866
- Published Date: Feb 10, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Medical Device Testing Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




