Global Closed System Transfer Devices Market
- An Outline of the Global Closed System Transfer Devices Market
- Market Definition and Segmentation
- Study Assumptions and Abbreviations
- Research Methodology & Approach
- Primary Research
- Secondary Research
- Data Triangulation
- SPSS Methodology
- Executive Summary
- Growth Drivers
- Major Roadblocks
- Opportunities
- Prevalent Trends
- Government Regulation
- Growth Outlook
- Competitive White Space Analysis – Identifying Untapped Market Gaps
- Risk Overview
- SWOT
- Technological Advancement
- Technology Maturity Matrix for Closed System Transfer Devices Market Recent News
- Regional Demand
- Global Closed System Transfer Devices by Geography – Strategic Comparative Analysis
- Strategic Segment Analysis: Closed System Transfer Devices Demand Landscape
- Closed System Transfer Devices Demand Trends Driven by rising cancer prevalence, increasing chemotherapy usage, and growing focus on healthcare worker safety and regulatory compliance (2026-2036)
- Root Cause Analysis (RCA) for discovering problems of the Closed System Transfer Devices Market
- Porter Five Forces
- PESTLE
- Comparative Positioning
- Global Closed System Transfer Devices – Key Player Analysis (2036)
- Competitive Landscape: Key Suppliers/Players
- Competitive Model: A Detailed Inside View for Investors
- Company Market Share, 2036 (%)
- Becton, Dickinson and Company (U.S.)
- CODAN Companies (Germany)
- CorMed (U.S.)
- Eraser Medikal Ltd. Şti (Turkey)
- EQUASHIELD (Israel)
- ICU Medical, Inc. (U.S.)
- JMS Co., Ltd. (Japan)
- Simplivia Healthcare Ltd. (Israel)
- Vygon (France)
- Yukon Medical, LLC (U.S.)
- Global Closed System Transfer Devices Market Outlook
- Market Overview
- Market Revenue by Value (USD Million), Volume (Million Tons), and Compound Annual Growth Rate (CAGR)
- Closed System Transfer Devices Market Segmentation Analysis (2026-2036)
- By Type
- Membrane-to-Membrane, Market Value (USD Million), and CAGR, 2026-2036F
- Needleless, Market Value (USD Million), and CAGR, 2026-2036F
- By Technology
- Diaphragm-based, Market Value (USD Million), and CAGR, 2026- 2036F
- Compartmentalized, Market Value (USD Million), and CAGR, 2026- 2036F
- Filtration, Market Value (USD Million), and CAGR, 2026-2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- By End user
- Hospitals and Clinics, Market Value (USD Million), and CAGR, 2026- 2036F
- Oncology Centers, Market Value (USD Million), and CAGR, 2026- 2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- Regional Synopsis, Value (USD Million), 2026-2036
- North America Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Europe Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Asia Pacific Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Latin America Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Middle East and Africa Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- By Type
- Market Overview
- North America Market
- Overview
- Market Value (USD Million), Current and Future Projections, 2026-2036
- Increment $ Opportunity Assessment, 2026-2036
- Segmentation (USD Million), 2026-2036, By
- By Type
- Membrane-to-Membrane, Market Value (USD Million), and CAGR, 2026-2036F
- Needleless, Market Value (USD Million), and CAGR, 2026-2036F
- By Technology
- Diaphragm-based, Market Value (USD Million), and CAGR, 2026- 2036F
- Compartmentalized, Market Value (USD Million), and CAGR, 2026- 2036F
- Filtration, Market Value (USD Million), and CAGR, 2026-2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- By End user
- Hospitals and Clinics, Market Value (USD Million), and CAGR, 2026- 2036F
- Oncology Centers, Market Value (USD Million), and CAGR, 2026- 2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- Country Level Analysis, Value (USD Million)
- S. Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Canada Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- By Type
- Overview
- Europe Market
- Overview
- Market Value (USD Million), Current and Future Projections, 2026-2036
- Increment $ Opportunity Assessment, 2026-2036
- Segmentation (USD Million), 2026-2036, By
- By Type
- Membrane-to-Membrane, Market Value (USD Million), and CAGR, 2026-2036F
- Needleless, Market Value (USD Million), and CAGR, 2026-2036F
- By Technology
- Diaphragm-based, Market Value (USD Million), and CAGR, 2026- 2036F
- Compartmentalized, Market Value (USD Million), and CAGR, 2026- 2036F
- Filtration, Market Value (USD Million), and CAGR, 2026-2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- By End user
- Hospitals and Clinics, Market Value (USD Million), and CAGR, 2026- 2036F
- Oncology Centers, Market Value (USD Million), and CAGR, 2026- 2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- Country Level Analysis, Value (USD Million)
- UK Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Germany Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- France Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Italy Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Spain Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Netherlands Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Russia Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Switzerland Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Poland Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Belgium Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Rest of Europe Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- By Type
- Overview
- Asia Pacific Market
- Overview
- Market Value (USD Million), Current and Future Projections, 2026-2036
- Increment $ Opportunity Assessment, 2026-2036
- Segmentation (USD Million), 2026-2036, By
- By Type
- Membrane-to-Membrane, Market Value (USD Million), and CAGR, 2026-2036F
- Needleless, Market Value (USD Million), and CAGR, 2026-2036F
- By Technology
- Diaphragm-based, Market Value (USD Million), and CAGR, 2026- 2036F
- Compartmentalized, Market Value (USD Million), and CAGR, 2026- 2036F
- Filtration, Market Value (USD Million), and CAGR, 2026-2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- By End user
- Hospitals and Clinics, Market Value (USD Million), and CAGR, 2026- 2036F
- Oncology Centers, Market Value (USD Million), and CAGR, 2026- 2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- Country Level Analysis, Value (USD Million)
- China Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Japan, Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- India Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- South Korea Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Australia Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Indonesia Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Malaysia Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Vietnam Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Thailand Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Singapore Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- New Zealand Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Rest of Asia Pacific Excluding Japan Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- By Type
- Overview
- Latin America Market
- Overview
- Market Value (USD Million), Current and Future Projections, 2026-2036
- Increment $ Opportunity Assessment, 2026-2036
- Year-on-Year Growth Forecast (%)
- Segmentation (USD Million), 2026-2036, By
- By Type
- Membrane-to-Membrane, Market Value (USD Million), and CAGR, 2026-2036F
- Needleless, Market Value (USD Million), and CAGR, 2026-2036F
- By Technology
- Diaphragm-based, Market Value (USD Million), and CAGR, 2026- 2036F
- Compartmentalized, Market Value (USD Million), and CAGR, 2026- 2036F
- Filtration, Market Value (USD Million), and CAGR, 2026-2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- By End user
- Hospitals and Clinics, Market Value (USD Million), and CAGR, 2026- 2036F
- Oncology Centers, Market Value (USD Million), and CAGR, 2026- 2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- Country Level Analysis, Value (USD Million)
- Brazil Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Argentina Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Mexico Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Rest of Latin America Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- By Type
- Overview
- Middle East & Africa Market
- Overview
- Market Value (USD Million), Current and Future Projections, 2026-2036
- Increment $ Opportunity Assessment, 2026-2036
- Year-on-Year Growth Forecast (%)
- Segmentation (USD Million), 2026-2036, By
- By Type
- Membrane-to-Membrane, Market Value (USD Million), and CAGR, 2026-2036F
- Needleless, Market Value (USD Million), and CAGR, 2026-2036F
- By Technology
- Diaphragm-based, Market Value (USD Million), and CAGR, 2026- 2036F
- Compartmentalized, Market Value (USD Million), and CAGR, 2026- 2036F
- Filtration, Market Value (USD Million), and CAGR, 2026-2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- By End user
- Hospitals and Clinics, Market Value (USD Million), and CAGR, 2026- 2036F
- Oncology Centers, Market Value (USD Million), and CAGR, 2026- 2036F
- Others, Market Value (USD Million), and CAGR, 2026-2036F
- Country Level Analysis, Value (USD Million)
- Saudi Arabia Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- UAE Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Israel Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Qatar Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Kuwait Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Oman Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- South Africa Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- Rest of Middle East & Africa Market Value (USD Million) and CAGR & Y-o-Y Growth Trend, 2026-2036F
- By Type
- Overview
- Global Economic Scenario
- World Economic Outlook
- About Research Nester
- Our Global Clientele
- We Serve Clients Across World
Closed System Transfer Devices Market Outlook:
Closed System Transfer Devices (CSTD) Market size is valued at USD 1.57 billion in 2025 and is projected to reach USD 7.07 billion by 2036, growing at a CAGR of 14.96% during the forecast period, i.e., 2026-2036. In 2026, the industry size of closed system transfer devices is evaluated at USD 1.75 billion.

The global CSTD market is expanding rapidly, driven by the urgent need to protect healthcare workers from hazardous drug exposure and the rising global cancer burden. According to the U.S. National Cancer Institute, in January 2022, the U.S. had approximately 18.1 million cancer survivors, a figure expected to grow to nearly 26 million by 2040, driven by improvements in early diagnosis and more effective treatment outcomes, fueling demand for chemotherapy and safe drug-handling technologies. The World Health Organization (WHO) reported nearly 9.7 million cancer deaths worldwide in 2022, with incidence projected to rise significantly, underscoring the importance of CSTDs in oncology care. Regulatory bodies such as NIOSH updated their 2022 guidelines recommending CSTDs as best practice for handling hazardous drugs, making compliance a strong growth driver. Hospitals and oncology clinics are increasingly investing in these devices to meet safety standards, reduce occupational risks, and improve patient outcomes. Post-pandemic healthcare infrastructure investments highlighted by UNCTAD also support the adoption of advanced protective technologies. Based on the UNCTAD World Investment Report 2025, international investment in SDG sectors like healthcare saw a +25% increase. Together, these factors position CSTDs as a critical technology in safeguarding healthcare workers and ensuring safe cancer treatment delivery worldwide.
Key Closed System Transfer Devices Market Insights Summary:
Regional Highlights:
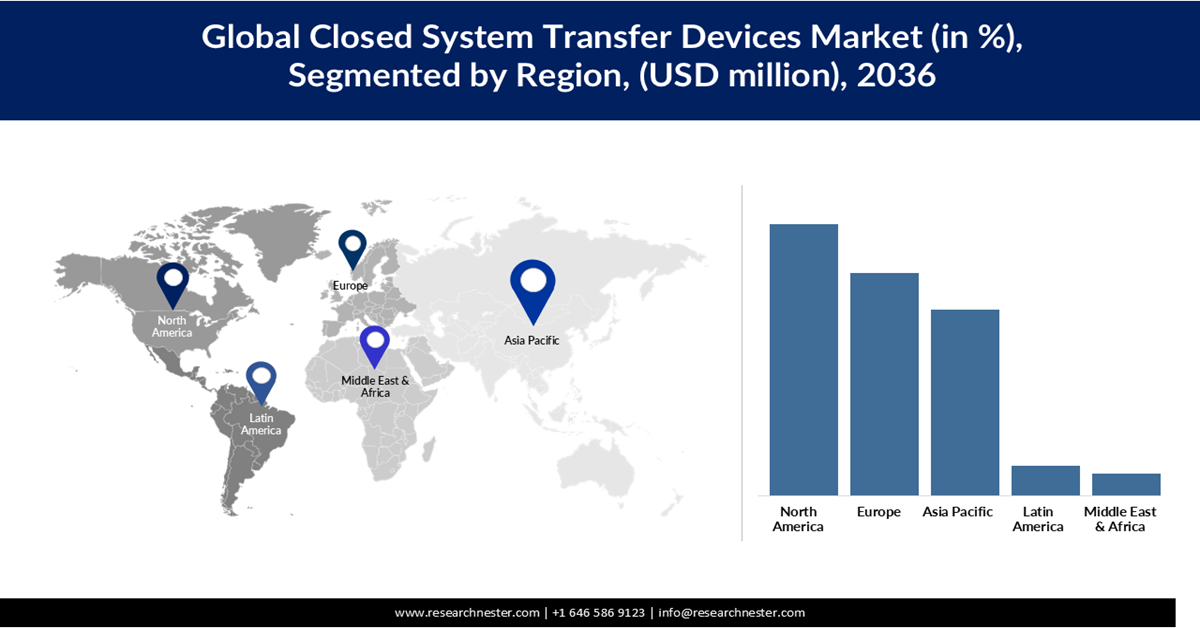
- The closed system transfer devices market in North America is projected to command a 37.08% share by 2036, reinforced by stringent safety regulations and increasing need to protect healthcare workers from hazardous drug exposure
- Asia Pacific is expected to witness notable growth with a 25.37% share by 2036, supported by rising cancer prevalence and expanding healthcare infrastructure
Segment Insights:
- The membrane-to-membrane segment in the closed system transfer devices market is projected to account for 62.66% share by 2036, driven by its highly secure drug transfer mechanism minimizing leakage and contamination risks
- The diaphragm-based segment is anticipated to hold a 50.96% share by 2036, propelled by its reliable barrier design ensuring safety against hazardous drug leakage
Key Growth Trends:
- Rising oncology drug volumes
- Growth in biologics
Major Challenges:
- Waste management and environmental concern
- Workflow complexity
Key Players: Becton, Dickinson and Company, CODAN Companies, CorMed, Eraser Medikal Ltd. Şti, EQUASHIELD, ICU Medical, Inc., JMS Co., Ltd., Simplivia Healthcare Ltd., Vygon, Yukon Medical, LLC.
Global Closed System Transfer Devices Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 1.57 billion
- 2026 Market Size: USD 1.75 billion
- Projected Market Size: USD 7.07 billion by 2036
- Growth Forecasts: 14.96% CAGR (2026-2036)
Key Regional Dynamics:
- Largest Region: North America (37.08% Share by 2036)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, United Kingdom, France
- Emerging Countries: China, India, South Korea, Brazil, Singapore
Last updated on : 1 May, 2026
Closed System Transfer Devices Market - Growth Drivers and Challenges
Growth Drivers
- Rising oncology drug volumes: The surge in oncology drug usage is a primary driver of CSTD adoption, as more chemotherapy agents are prepared and administered in healthcare settings. According to the CDC’s U.S. Cancer Statistics, there were 1,851,238 new cancer cases reported in 2022 and 613,349 cancer deaths in 2023, figures that highlight the scale of chemotherapy demand. With cancer incidence rising globally, oncology drugs are increasingly handled in hospitals and clinics, elevating the risk of occupational exposure. CSTDs mitigate this risk by preventing leaks and contamination during drug transfer, making them essential for oncology departments. The growing patient pool directly translates into higher drug volumes, which in turn accelerates the adoption of CSTDs as standard safety equipment.
- Growth in biologics: Biologics, including monoclonal antibodies and advanced therapies, are expanding rapidly and often require specialized handling due to their complexity and potential hazards. The Biotechnology Innovation Organization (BIO) reported that the U.S. bioscience industry generated over USD 3.2 trillion in economic output in 2023 and employed nearly 2.3 million Americans, reflecting the sector’s rapid growth. As biologics become central to cancer treatment and other chronic diseases, healthcare facilities face increased handling of sensitive and potentially hazardous compounds. CSTDs provide the necessary containment to ensure biologics are safely prepared and administered, protecting both healthcare workers and patients. The expansion of biologics broadens the scope of hazardous drugs beyond traditional chemotherapy, thereby driving demand for CSTDs across multiple therapeutic areas.
- Regulatory enforcement intensifies adoption: Regulatory bodies are increasingly mandating the use of CSTDs to safeguard healthcare workers. These updates highlight the growing recognition of occupational risks and the need for protective technologies. Enforcement of guidelines by NIOSH, OSHA, and USP compels hospitals and pharmacies to adopt CSTDs to remain compliant, avoid penalties, and reduce liability. This regulatory push transforms CSTDs from optional safety tools into mandatory equipment, ensuring widespread closed system transfer devices market adoption. As enforcement intensifies, healthcare providers are investing in CSTDs not only for compliance but also to demonstrate commitment to worker safety and patient care.
Challenges
- Waste management and environmental concern: CSTDs often involve single-use components, which generate significant amounts of medical plastic waste. Healthcare facilities must balance worker safety with environmental responsibility, and the rising scrutiny from agencies like the U.S. Environmental Protection Agency on medical waste disposal adds pressure. The additional costs and complexities of safe disposal can discourage widespread adoption, particularly in regions with strict environmental regulations. This tension between safety and sustainability slows down market penetration despite the clear protective benefits of CSTDs.
- Workflow complexity: Another restraint is the added workflow complexity that CSTDs introduce into clinical practice. While they enhance safety, their use requires additional training, changes in drug preparation protocols, and sometimes longer handling times. According to the NIOSH guidelines on hazardous drug handling, proper implementation of CSTDs involves strict adherence to protocols and staff education, which can be resource-intensive. Hospitals and pharmacies may hesitate to adopt CSTDs due to concerns about disrupting established workflows, increasing operational costs, and requiring ongoing staff training. This complexity can act as a barrier, especially in smaller healthcare facilities with limited resources, slowing down the pace of closed system transfer devices (CSTD) market growth.
Closed System Transfer Devices Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2036 |
|
CAGR |
14.96% |
|
Base Year Market Size (2025) |
USD 1.57 billion |
|
Forecast Year Market Size (2036) |
USD 7.07 billion |
|
Regional Scope |
|
Closed System Transfer Devices Market Segmentation:
Type Segment Analysis
The membrane-to-membrane segment is projected to account for 62.66% of the closed system transfer devices market share by 2036. This growth is driven by its highly secure drug transfer mechanism, which significantly minimizes the risks of leakage and contamination. Unlike needle-based systems, membrane-to-membrane designs create a sealed barrier during drug preparation and administration, which aligns with NIOSH recommendations for minimizing occupational exposure to hazardous drugs. This reliability makes them especially valuable in oncology, where large volumes of chemotherapy are handled. Beyond cancer, the rising use of biologics also contributes. In 2022, small- to mid-sized biopharma companies played a dominant role in innovation, contributing 65% (24 out of 37) of new drug approvals, while large biopharma companies accounted for just 35% (13 approvals). Notably, these smaller firms were responsible for 16 small-molecule drug approvals and eight biologic approvals, underscoring their growing impact across both therapeutic categories. As more complex therapies enter clinical practice, healthcare facilities increasingly prefer membrane-to-membrane CSTDs for their proven safety and compliance advantages. This combination of regulatory support, growing oncology volumes, and biologics expansion positions the segment as a major driver of overall closed system transfer devices (CSTD) market growth.
Technology Segment Analysis
The diaphragm-based segment is expected to dominate the closed system transfer devices market with a share of 50.96% by 2036, as it provides a highly reliable barrier against hazardous drug leakage, ensuring safety during preparation and administration. This design uses a flexible diaphragm that reseals after each transfer, reducing the risk of aerosolization or accidental spills. According to NIOSH, healthcare workers are at risk of exposure to over 200 hazardous drugs listed in the 2024 update, making protective technologies like diaphragm-based CSTDs essential. Their durability and ease of repeated use make them particularly attractive in high-volume oncology and hospital pharmacy settings. Additionally, the growing use of biologics further supports adoption, as diaphragm systems can safely handle complex therapies. By combining regulatory compliance, worker safety, and operational efficiency, the diaphragm-based segment is emerging as a key contributor to overall closed system transfer devices (CSTD) market expansion.
End user Segment Analysis
The hospitals and clinics segment is expected to reach USD 5,105.73 million with a CAGR of 14.90% between 2026 and 2036. Hospitals and clinics handle the largest volumes of hazardous drugs, particularly chemotherapy and biologics. Their role as primary treatment centers for cancer and chronic diseases makes them highly dependent on safe drug-handling technologies. Hospitals and clinics also face strict compliance requirements that mandate protective measures to safeguard healthcare workers. By integrating CSTDs into daily workflows, these institutions not only ensure regulatory compliance but also enhance patient and staff safety. The combination of high patient throughput, regulatory obligations, and focus on occupational health positions hospitals and clinics as the dominant end-user segment, fueling consistent demand and closed system transfer devices market expansion.
Our in-depth analysis of the closed system transfer devices includes the following segments:
|
Segments |
Subsegments |
|
Type |
|
|
Technology |
|
|
End user |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Closed System Transfer Devices Market - Regional Analysis
North America Market Insights
The closed system transfer devices market in North America is expected to hold a share of 37.08% by 2036. The region is witnessing steady growth, largely driven by stringent safety regulations and the rising need to protect healthcare workers from hazardous drug exposure. Hospitals, oncology centers, and infusion clinics are increasingly adopting these devices to comply with standards. Innovation in design and broader applications beyond oncology are also fueling demand. While high costs remain a challenge, the emphasis on workplace safety and expanding use cases continues to strengthen market momentum.
The U.S. closed system transfer devices market is expanding rapidly, supported by USP <800> compliance requirements and NIOSH guidelines that mandate CSTDs as engineering controls for hazardous drug handling. According to the CDC/NIOSH, about 8 million U.S. healthcare workers are potentially exposed to hazardous drugs, including nurses, pharmacists, and operating room staff, with risks ranging from infertility and miscarriages to cancers. This large at-risk workforce has accelerated adoption in hospitals, oncology centers, and infusion clinics. The FDA has approved CSTDs since the late 1990s, and innovation continues to improve compatibility with biologics and specialty drugs. Overall, the U.S. market is characterized by strong regulatory enforcement, widespread adoption in hospitals, and steady expansion into outpatient and specialty care settings.
In Canada, the CSTD market is growing at a moderate but consistent pace, supported by occupational health standards that align with NIOSH and USP guidelines. Provincial health authorities encourage adoption in oncology centers and large hospitals, particularly where cancer treatment volumes are high. Healthcare worker safety is a priority, with CSTDs recognized as effective in reducing exposure to cytotoxic drugs that pose reproductive and carcinogenic risks. Research collaborations and pilot programs across Canadian institutions are helping expand awareness and implementation. While adoption varies by province, the overall trend points toward increasing integration of CSTDs into hazardous drug handling practices.
Europe Market Insights
The Europe CSTD market is steadily growing, driven by stricter EU occupational safety regulations, rising cancer prevalence, and increasing awareness of healthcare worker protection. Hospitals and oncology centers are leading adopters, while biologics and specialty injectables are expanding the scope of CSTDs. Despite fragmented regulations and pricing pressures in public healthcare systems, innovation and harmonization of safety standards are supporting broader adoption.
The UK market for CSTDs is expanding as hospitals and oncology centers adopt these devices to comply with occupational safety standards. Healthcare workers face risks from hazardous drug exposure, making engineering controls such as CSTDs increasingly important for reducing occupational hazards. The NHS has been gradually integrating CSTDs into oncology and pharmacy workflows, particularly in large teaching hospitals. Growing use of biologics and chemotherapy drugs further accelerates demand. Government-backed safety initiatives continue to ensure adoption across the healthcare system.
The closed system transfer devices market in Germany is growing steadily, supported by the country’s strong oncology infrastructure, rising cancer burden, and expanding medical technology sector. Germany remains the largest medical technology market in Europe, with total medtech sales reaching approximately EUR 43 billion in 2023, which reflects increasing adoption of advanced safety devices such as CSTDs. A key driver is the high and growing volume of cancer treatment; over 1.45 million cancer patients were treated in German hospitals in 2024, significantly increasing the need for safe chemotherapy handling systems. Expanding cancer care ecosystem, combined with strict occupational safety practices in Germany’s healthcare system, is driving sustained demand for CSTDs, positioning the country as a key growth closed system transfer devices market within Europe.
Asia Pacific Market Insights
Asia Pacific CSTD market is projected to grow at a market share of 25.37% by 2036, driven by rising cancer prevalence, expanding healthcare infrastructure, and increasing awareness of healthcare worker safety. Countries across the region are adopting CSTDs to reduce occupational exposure to hazardous drugs, with oncology centers and large hospitals leading the way. Regulatory alignment and investment in biologics are further supporting adoption. Despite cost challenges, the emphasis on worker protection ensures continued growth.
China CSTD market is expanding as the country invests heavily in oncology care and worker safety. Hospitals and cancer treatment centers are increasingly adopting CSTDs to protect pharmacists and nurses from hazardous drug exposure. According to ILOSTAT, China reported millions of non-fatal occupational injuries annually, underscoring the importance of engineering controls in healthcare settings. According to the WHO, rising cancer incidence also drives demand, with China recording over 4.8 million new cancer cases in 2022. Government-backed initiatives to modernize hospital infrastructure and improve occupational safety standards are accelerating adoption. Overall, China’s large patient base and regulatory focus make it a key growth hub for CSTDs in Asia Pacific.
India CSTD market is witnessing steady growth, driven by the rising burden of cancer and increasing awareness around the safety of healthcare professionals handling hazardous drugs. Adoption is primarily concentrated in oncology centers and large tertiary hospitals, especially across major metropolitan cities, where advanced treatment infrastructure and stricter safety protocols are more prevalent. According to ILOSTAT, India reported hundreds of thousands of non-fatal occupational injuries annually, highlighting the scale of workplace risks across sectors, including healthcare. Government initiatives to strengthen oncology infrastructure and occupational safety standards are supporting adoption, though cost remains a barrier in smaller facilities. As awareness grows, India is expected to see broader integration of CSTDs across its healthcare system.
Key Closed System Transfer Devices Market Players:
- Becton, Dickinson and Company (U.S.)
- CODAN Companies (Germany)
- CorMed (U.S.)
- Eraser Medikal Ltd. Şti (Turkey)
- EQUASHIELD (Israel)
- ICU Medical, Inc. (U.S.)
- JMS Co., Ltd. (Japan)
- Simplivia Healthcare Ltd. (Israel)
- Vygon (France)
- Yukon Medical, LLC (U.S.)
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- A global leader, BD dominates the closed system transfer devices (CSTD) market with a strong portfolio including systems like PhaSeal. It benefits from a vast global distribution network and high brand trust in hospital settings. The company invests heavily in R&D and regulatory compliance to maintain leadership. Its scale and innovation capabilities allow it to set industry benchmarks.
- ICU Medical is a major player known for its needleless CSTD technologies such as ChemoClave and ChemoLock. The firm emphasizes safety, ease of use, and contamination prevention in drug handling. It has strengthened its position through acquisitions and product innovation. Its solutions are widely adopted in oncology and infusion therapy.
- Equashield specializes in advanced membrane-to-membrane CSTD systems designed to minimize drug exposure. The company is recognized for its closed syringe technology and continuous product upgrades. It has expanded globally through partnerships and distribution networks. Its focus on safety and performance has made it a strong competitor in premium segments.
- B. Braun offers a broad portfolio of drug delivery and CSTD solutions, with strong presence in Europe and emerging markets. The company leverages its healthcare ecosystem and manufacturing expertise for market penetration. It emphasizes product reliability, safety, and compliance with global standards. Strategic expansion and diversification strengthen its competitive position.
- Corvida Medical is an emerging innovator focusing on next-generation CSTDs with improved usability and safety features. Though smaller than industry leaders, it competes through specialized product design and innovation. The company targets niche and evolving healthcare needs. Its growth reflects increasing opportunities for new entrants in the closed system transfer devices market.
Below is the list of the key players operating in the global closed system transfer devices market:
Key players such as Becton, Dickinson and Company, ICU Medical, and B. Braun Melsungen AG are driving the global closed system transfer devices (CSTD) market through continuous innovation in safer, needle-free, and more efficient drug transfer technologies. They are expanding their market presence via strategic partnerships, acquisitions, and entry into emerging regions, while also ensuring compliance with stringent healthcare safety regulations. At the same time, strong investments in research and development are enabling the launch of advanced, user-friendly products with improved compatibility, which is accelerating adoption across hospitals and oncology care settings worldwide.
Corporate Landscape of the Global Closed System Transfer Devices Market:
Recent Developments
- In September 2025, ICU Medical, Inc., a global medical technology company specializing in IV therapy and critical care, announced that it had received new 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Clave portfolio of needle-free connectors. This clearance introduces a new labeling claim based on peer‑reviewed study data, showing that acute care hospitals using Clave connectors at high volumes experienced a 19% lower relative risk of central line‑associated bloodstream infections (CLABSI). The findings also highlight reduced healthcare costs and improved patient survival rates. With this milestone, ICU Medical strengthens its commitment to advancing patient safety and infection prevention in critical care environments.
- In December 2025, B. Braun Medical Inc., a leader in smart infusion therapy and pain management, announced the launch of Piperacillin and Tazobactam, one of the most widely used injectable antibiotics in the U.S., for use in its DUPLEX® Drug Delivery System. The product, Piperacillin and Tazobactam for Injection and Sodium Chloride Injection, is available in three strengths: 2.25 g/50 mL, 3.375 g/50 mL, and 4.5 g/100 mL. This introduction highlights B. Braun’s commitment to expanding access to essential antibiotics while enhancing safety, efficiency, and reliability in drug delivery.
- Report ID: 4090
- Published Date: May 01, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




