Thrombectomy Devices Market Outlook:
Thrombectomy Devices Market size was USD 2.2 billion in 2025 and is projected to reach USD 4.2 billion by the end of 2035, increasing at a CAGR of 7.5% during the forecast period, i.e., 2026-2035. In 2026, the industry size of thrombectomy devices is assessed at USD 2.3 billion.

The worldwide thrombectomy devices market is gaining increased exposure, owing to a surge in cardiovascular disease and stroke burden, severe clinical results through pharmacological therapies, an increase in the aging population, and an epidemic in atrial fibrillation. In this regard, as per an article published by NLM in January 2025, stroke is the second leading cause of death, affecting 7 million people globally. Additionally, there has been a surge in stroke incidents over the past 4 years by 70%, with 44% deaths from stroke, with the majority of the burden residing in developing nations. Besides, the approximate international stroke expense is more than USD 890 billion, which accounts for 0.6% of gross domestic product (GDP).
Moreover, the minimally invasive surgery (MIS) integration and AI-powered devices adoption are also positively fueling the market growth globally. According to a report published by NLM in January 2025, a clinical study was conducted on 408 hospitals, of which 153 (38%) implemented robotic-assisted surgery (RAS) for conducting general surgeries. Based on this, the standard MIS rate for hospitals adopting RAS in comparison to hospitals not adopting was 1.0% prior to the index rate of 1.1%. This indicated an increase in the MIS rate among hospitals integrating MIS across patient gender, age, race, ethnicity, and payer, thereby suitable for the market’s upliftment.
Key Thrombectomy Devices Market Insights Summary:
Regional Highlights:
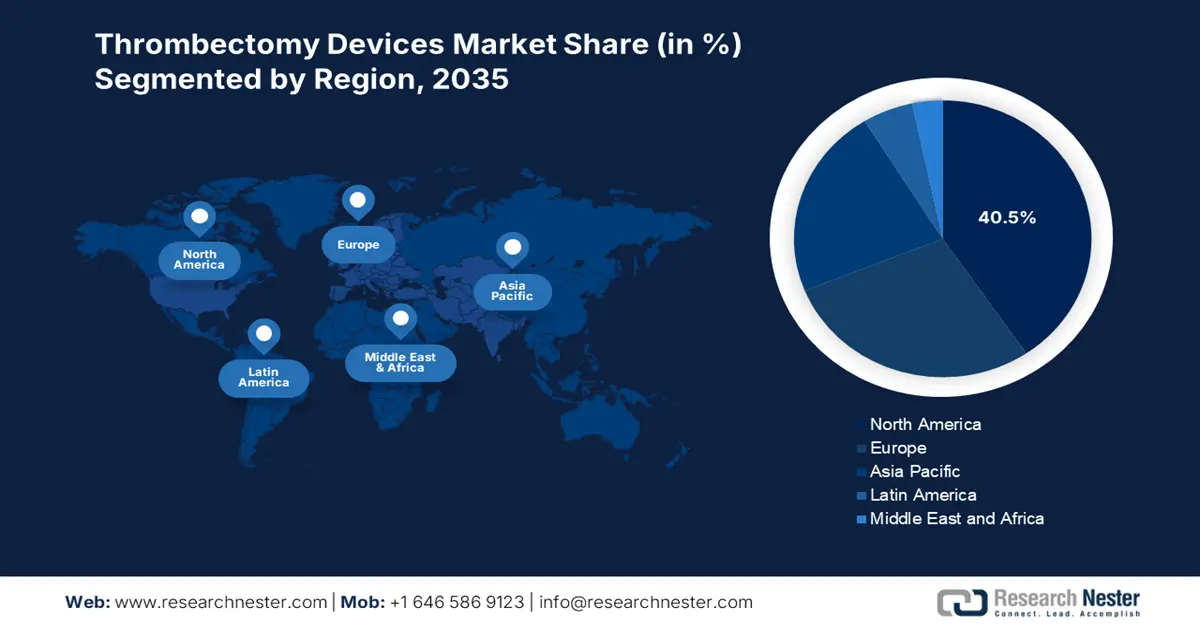
- North America is anticipated to command a 40.5% share by 2035 in the thrombectomy devices market, supported by harmonized regulatory frameworks, integrated supply chains, and increased clinical clearances such as AVENTUS 510(k) approval.
- Asia Pacific is projected to be the fastest-growing region through 2026–2035, reinforced by its disproportionate stroke burden, diverse healthcare capacities, and varying regulatory pathways.
Segment Insights:
- The Hospitals segment in the thrombectomy devices market is forecast to achieve a 58.7% share by 2035, stimulated by continuous emergency readiness, extensive certifications, multidisciplinary expertise, and reimbursement policies encouraging the adoption of advanced aspiration systems and stent retrievers.
- The mechanical thrombectomy devices segment is set to represent the second-largest share by 2035, strengthened by its proficiency in removing large clots and improving acute ischemic stroke outcomes.
Key Growth Trends:
- Rise in vascular diseases burden
- Expansion in treatment indications
Major Challenges:
- Strict Health technology assessment and affordable obstacles
- Reference pricing and capping mechanisms
Key Players: Medtronic plc (Ireland), Stryker Corporation (U.S.), Johnson & Johnson (DePuy Synthes) (U.S.), Penumbra, Inc. (U.S.), Boston Scientific Corporation (U.S.), Abbott Laboratories (U.S.), Spectranetics (Philips) (Netherlands), Inari Medical, Inc. (U.S.), BD (Becton, Dickinson and Company) (U.S.), Acandis GmbH (Germany), Phenox GmbH (Germany), Imperative Care, Inc. (U.S.), Rapid Medical (Israel), Vesalio (U.S.), Lemaitre Vascular, Inc. (U.S.).
Global Thrombectomy Devices Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 2.2 billion
- 2026 Market Size: USD 2.3 billion
- Projected Market Size: USD 4.2 billion by 2035
- Growth Forecasts: 7.5% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (40.5% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, China, Germany, Japan, United Kingdom
- Emerging Countries: India, Brazil, South Korea, Mexico, Turkey
Last updated on : 29 August, 2025
Thrombectomy Devices Market - Growth Drivers and Challenges
Growth Drivers
- Rise in vascular diseases burden: These particular disorders signify global health challenges, substantially contributing to disability, mortality, and healthcare expenses, which in turn, is uplifting the thrombectomy devices market demand internationally. As per an article published by NLM in February 2025, age-standardized estimates for vascular diseases account for a quarter, which is 24.8% of overall deaths, especially in India. In addition, the age-standardized heart disease death rate of 272 per 100,000 population is more than the worldwide average of 235 per 100,000 population, thereby enhancing the market’s exposure.
- Expansion in treatment indications: These are essential in providing an evidence-based and rational foundation for undertaking treatment decisions to ensure suitable and beneficial interventions, thus suitable for uplifting the thrombectomy devices market. According to an article published by the World Health Organization in March 2025, successfully scaling up primary healthcare (PHC) interventions in low and middle-income nations possesses the capability to save 60 million lives and enhance average life expectancy by 3.7 years by the end of 2030. Additionally, approximately 75% of health gains can be achieved from the Sustainable Development Goals, thereby denoting a huge scope for the market to flourish.
- Advancements in device technologies: This effectively optimizes patient diagnostics, treatments, care, along with management of conditions, thereby boosting the thrombectomy device market’s exposure. As stated in the October 2022 NLM report, 5% of global patients account for 50% of all expenses, along with a surge in chronic diseases demanding continuous treatment. Based on this, machine learning is one such technology that can be implemented to carefully recognize patients being more prone to develop common illnesses, which in turn, denotes a positive impact on the market globally.
2023 Risk Factors Associated with Cardiovascular Diseases Driving the market
|
Risk Factors |
Prevalence |
|
Hypertension |
24% |
|
Diabetes |
10% |
|
Dyslipidemia |
39% |
|
Smoking |
36% |
|
Alcohol Consumption |
16% |
|
Tobacco utilization |
1.0 million deaths |
|
Improper physical activity |
11% |
|
Poor Diet quality |
12% |
|
Life events |
32% |
Source: NLM, November 2025
Challenges
- Strict Health technology assessment and affordable obstacles: Beyond simplified regulatory acceptance for efficacy and safety, manufacturers need to navigate effective HTA processes to demand cost-effective evidence. Agencies, including NICE in the UK and Germany’s G-BA, readily demand extended dossiers for successfully comparing devices’ long-lasting care standards. This has readily necessitated expensive outcomes and real-world proof generation, frequently taking 12 to 24 months for clearance. Besides, a device can be clinically superior, but often fails to achieve reimbursement due to an unjustified price premium, which is negatively impacting the market globally.
- Reference pricing and capping mechanisms: Different national health systems have employed International Reference Pricing (IRP), thereby setting a maximum reimbursable price for devices based on the average pricing system from different nations. This practice has been utilized across Canada and Europe, which has readily crushed profit margins and disincentivized innovation, thus causing a hindrance in the market internationally. However, manufacturers must tactically unveil across high-value-based markets before getting entry in nations with stringent pricing strategies, frequently accepting low margins.
Thrombectomy Devices Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
7.5% |
|
Base Year Market Size (2025) |
USD 2.2 billion |
|
Forecast Year Market Size (2035) |
USD 4.2 billion |
|
Regional Scope |
|
Thrombectomy Devices Market Segmentation:
End user Segment Analysis
Hospitals segment in the thrombectomy devices market is anticipated to garner the highest share of 58.7% by the end of 2035. The segment’s upliftment is highly driven by its 24/7 emergency capability, wide-ranging stroke cancer certifications, and the presence of multidisciplinary teams. Besides, high surgical procedure volumes are highly favorable for CAM reimbursement policies, particularly in the U.S., and similar reforms across other nations, which have readily incentivized the implementation of progressive technologies, including aspiration systems and stent retrievers, thereby suitable for the segment’s development internationally.
Product Type Segment Analysis
The mechanical thrombectomy devices segment is projected to hold the second-highest market share by the end of the forecast timeline. The segment’s growth is driven by its ability to effectively restore blood flow by carefully removing large blood clots and enhancing patient outcomes for acute ischemic stroke. As per an article published by NLM in August 2023, approximately 10% of patients are readily suitable for mechanical thrombectomy, of which only 3% of patients from the UK are presently offered this treatment, thus suitable for the segment’s growth in the market.
Application Segment Analysis
Neurovascular thrombectomy segment in the market is expected to account for the third-highest share during the projected period. The segment’s growth is highly fueled by its increased effectiveness, minimally invasive treatment for removing blood clots from the brain artery, and successfully diminishing disability by optimizing the quality of life. As per the April 2022 NLM report, a clinical study was conducted on 74 patients regarding the advanced neurovascular access (ANA) thrombectomy system application. The result indicated 55.6% as the recanalization rate, along with 57.5% as the functional outcome, thereby suitable for the segment’s upliftment.
Our in-depth analysis of the global market includes the following segments:
|
Segment |
Subsegments |
|
End user |
|
|
Product Type |
|
|
Application |
|
|
Technology |
|
|
Utility |
|
|
Clot Type |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Thrombectomy Devices Market - Regional Analysis
North America Market Insights
North America is anticipated to be the dominating region by account for the largest market share of 40.5% by the end of 2035. The market’s growth in the region is propelled by harmonized regulatory policies, integrated supply chains, cross-border clinical studies, and the presence of notable players. For instance, in June 2025, Inquis Medical’s AVENTUS Thrombectomy System has successfully achieved the U.S. Food and Drug Administration (FDA) 510(k) clearance. The purpose of this was to extend the indications for effectively aiding pulmonary embolism, thereby suitable for the market’s upliftment.
U.S. is growing significantly, owing to the presence of the CMS Inpatient Prospective Payment System (IPPS), consolidated hospitals, increased prevalence of comorbidities, as well as a high litigation environment. According to a report published by AHA Journals in November 2023, almost half of the population in the country is projected to be obese, which includes 1 in 4 people being severely affected with this condition by the end of 2030. Besides, 78% of hypertension among men and 65% among women is readily attributable to obesity, thus enhancing the market’s demand in the country.
The thrombectomy devices market in Canada is also growing due to the existence of provincial health technology assessment (HTA), single-payer system availability, the presence of concentrated urban centers, and provincial bulk purchasing. As stated in the Obesity Canada November 2024 report, the inaction expense of successfully aiding obesity in the country has reached USD 27.6 billion, which accounts for almost 20% than the previous estimation. In addition, with nearly 1 in 3 people residing with this condition, both direct and indirect expenses are staggering.
Obesity Incidence in North America
|
U.S. |
Incidence |
Canada |
Incidence |
|
Early 20s |
30.5% |
2021 |
65% |
|
2017-2020 |
41.9% |
Men versus Women |
39.7% and 31.3% |
|
Approximate Medical Expense |
USD 173 billion |
18 to 34 years |
22.5% |
|
2035 prediction |
More than ½ of the population |
More than 80 years |
21.2% |
|
- |
- |
Low versus High Household Income |
30.8% and 26.6% |
|
- |
- |
Rural versus Urban |
33.6% and 28.6% |
Sources: Obesity Medicine Organization, June 2024; Government of Canada, June 2025
APAC Market Insights
Asia Pacific market is projected to be the fastest-growing region during the forecast period. The market’s growth in the region is subject to disproportionate disease burden of stroke cases, extreme economic diversity, rapid development of facilities for aiding stroke, and diversified regulatory pathways. As per the April 2024 NLM article, an estimated 9.5 to 10.6 million stroke cases will affect the region every year. Besides, the mortality rate in Singapore is moderate, accounting for 47.9% per 100,000 person-years, and highest in Indonesia, with 193.3% per 100,000 person-years, thus suitable for the market’s growth.
The thrombectomy devices market in China is gaining increased traction, owing to the existence of the centralized Healthy China 2030 plan, the presence of sheer patient volume, the NMPA’s fast-track clearance for regional innovation, and the availability of the tiered hospital system. According to an article published by NLM in April 2022, over 2 million patients in the country have utilized outpatient services, particularly at the Peking University First Hospital in each year. Based on this, it has been noted that the average growth rate for outpatients has been 16.6%, with the utilization rate being 54.1%, thereby denoting the market’s requirement in the country.
India market is also growing due to extreme price sensitivity, an effective shortage of trained specialists, fragmentation in public health funding, and an increase in the young stroke population. As stated in the February 2022 NLM article, the overall neurological diseases caused by stroke have been determined to be 37.9%, with a 95% uncertainty level. In addition, between 20 to 64 years of age, there has been a surge in stroke deaths by almost 36.7%. Therefore, with all these incidents, there is a huge opportunity for the market to flourish in the country.
Europe Market Insights
Europe market is expected to account for a considerable share by the end of the projected timeline. The market’s upliftment in the region is highly fueled by centralized regional MDR acceptance, the presence of the EU4Health programme support, domestic clinical guidelines availability, and price preference between markets. As per the March 2023 NLM article, health technology is an essential industry for the Finnish economy, and its exports over the past 5 years totaled to almost EUR 2.5 billion. In addition, the overall healthcare in the country has been effectively digitalized in both private and public sectors, which is positively contributing towards the market’s growth in the overall region.
Germany market is steadily growing, owing to the existence of the innovative G-BA process, compulsory stroke unit certification, diagnosis-based group system, and a rapid increase in the aging population. According to the 2023 OECD report, circulatory disorders in the country accounted for more than 33% of deaths in 2021. In addition, cancer constitutes 22% of overall deaths, with lung cancer being the most common cause of death in the country, thus denoting a positive outlook for enhancing the market’s dominance in the overall country, which in turn has increased export and import of catheters as well.
The thrombectomy devices market in France is also developing due to the presence of a top-down national stroke strategy, mission-based hospital funding, centralized facility, and stringent cost-effective demands. As stated in the May 2025 NLM article, there has been an increase in specialists, especially in the country’s private sector, by 57.4% as of 2022, in comparison to 38.0% in previous years. Besides, the majority of funding in the domestic healthcare is contributed by employee and employer payroll, accounting for 33%, 24% of general social contributions, and 20% of other taxes.
Needles, Catheters, and Cannula 2023 Export and Import in Europe
|
Countries |
Export |
Import |
|
Ireland |
USD 4.4 billion |
USD 747 million |
|
Germany |
USD 1.8 billion |
USD 2.7 billion |
|
Netherlands |
USD 2.9 billion |
USD 4.9 billion |
|
Belgium |
USD 953 million |
USD 1.2 billion |
|
Hungary |
USD 670 million |
USD 223 million |
|
France |
USD 467 million |
USD 1.2 billion |
Source: OEC, July 2025
Key Thrombectomy Devices Market Players:
- Medtronic plc (Ireland)
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Stryker Corporation (U.S.)
- Johnson & Johnson (DePuy Synthes) (U.S.)
- Penumbra, Inc. (U.S.)
- Boston Scientific Corporation (U.S.)
- Abbott Laboratories (U.S.)
- Spectranetics (Philips) (Netherlands)
- Inari Medical, Inc. (U.S.)
- BD (Becton, Dickinson and Company) (U.S.)
- Acandis GmbH (Germany)
- Phenox GmbH (Germany)
- Imperative Care, Inc. (U.S.)
- Rapid Medical (Israel)
- Vesalio (U.S.)
- Lemaitre Vascular, Inc. (U.S.)
The international thrombectomy devices market is severely consolidated and effectively dominated by large-scale Europe and U.S.-based medtech giants. Notable key players, including Johnson & Johnson, Stryker, and Medtronic, have leveraged extensive R&D activities and worldwide commercial networks to maintain dominance through continued clinical evidence and product iteration generation. The ultimate focus is on aspiration thrombectomy, AI-based implementation, and robotic-assisted systems. Besides, to extend accessibility, notable organizations have readily pursued value-specific pricing deals, along with physician training programs, thereby suitable for uplifting the market.
Here is a list of key players operating in the global market:
Recent Developments
- In October 2024, Royal Philips and Medtronic Neurovascular jointly announced a tactical advocacy partnership by effectively delivering stroke care services to save lives and diminish long-lasting disability among patients.
- In September 2024, FlowPhysix Inc. declared its strategic partnership with 3comma Medical for serving as the global commercial partner for the FLOWRUNNER Aspiration System.
- Report ID: 8029
- Published Date: Aug 29, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Thrombectomy Devices Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




