RNA Based Therapeutics Market Outlook:
RNA Based Therapeutics Market size was USD 8.4 billion in 2025 and is anticipated to reach USD 26.2 billion by the end of 2035, increasing at a CAGR of 13.5% during the forecast period, i.e., 2026-2035. In 2026, the industry size of RNA based therapeutics is assessed at USD 9.5 billion.

The worldwide market’s growth is highly driven by the deployment and development of RNA-based vaccines, which have displayed rapid upliftment and increased efficacy. In addition, mRNA‐based approaches, CRISPR‐Cas‐mediated genome editing, and RNAi‐based mechanisms are a few novel therapeutics that positively impact the overall market growth. In this regard, an article published by The Embo Journal in September 2023 denoted that mRNA vaccines displayed a 90% efficacy rate through clinical trials to protect pathogens, including rabies virus, influenza A virus, respiratory syncytial virus (RSV), and Zika virus, thereby suitable for boosting the market globally.
Moreover, the aspect of clotting and metabolic disorders, along with low lipoprotein as part of cardiovascular diseases (CVD), is also uplifting the market demand. According to an article published by the World Health Organization (WHO) in 2025, CVD has become one of the leading causes of death, which includes 17.9 million deaths every year. Additionally, over 4 out of 5 deaths occur due to heart strokes and attacks, and 1/3rd of the deaths prematurely take place among people within 70 years of age. However, antisense oligonucleotides (ASO) as an RNA therapy have been useful for evaluation. Based on this, the December 2023 Non-coding RNA Research article denoted the evaluation of 55% for hypercholesterolemia, 20% for transthyretin amyloidosis, along with atrial fibrillation and atherosclerotic cardiovascular disease (ASCVD), thus suitable for the market growth.
Key RNA Based Therapeutics Market Insights Summary:
Regional Highlights:
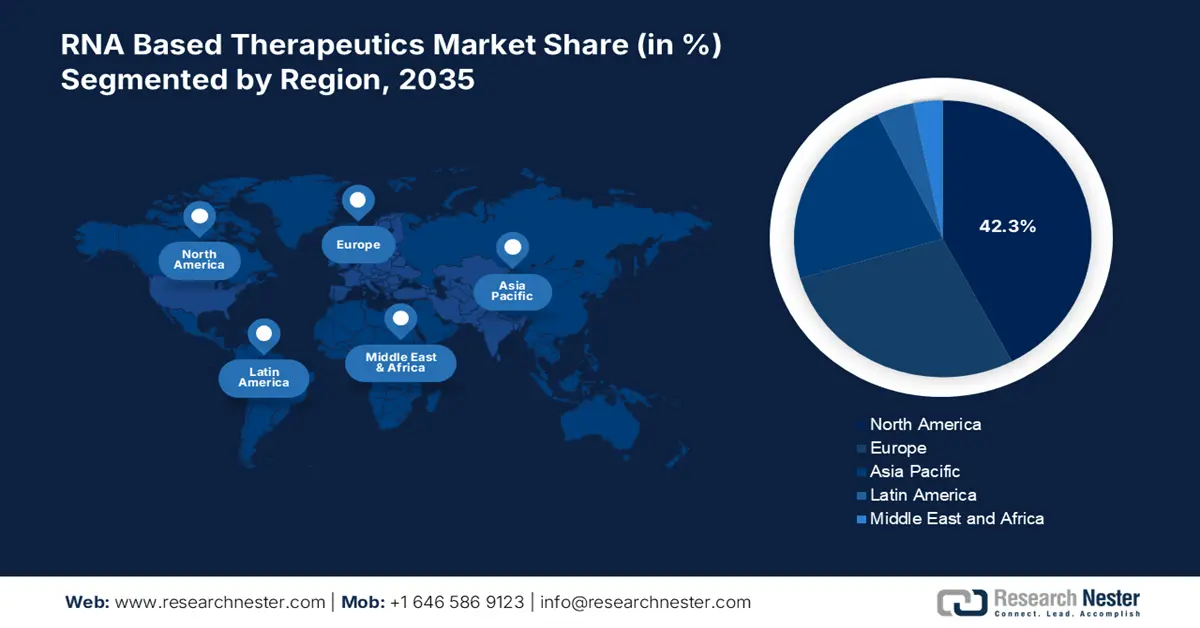
- By 2035, North America in the RNA based therapeutics market is predicted to command a 42.3% share, sustained by ongoing R&D activities, administrative advantages, increased healthcare expenditure, and strong partnerships.
- By 2035, Asia Pacific is forecast to capture a 22.4% share, elevated by government-driven strategies, pharmaceutical investments, and administrative clearances.
Segment Insights:
- By 2035, the biopharmaceutical companies segment in the RNA based therapeutics market is projected to achieve a 58.6% share, spurred by increasing drug development, advanced therapy delivery, and expanding adoption of artificial intelligence.
- By 2035, the mRNA therapeutics segment is anticipated to hold a 48.5% share, propelled by the strategic emphasis on next-generation vaccine platforms and cancer immunotherapies suitable for mRNA infectious diseases.
Key Growth Trends:
- Rise in chronic diseases
- Surge in research and investment
Major Challenges:
- Fragmentation in regulation
- Storage and cold chain expenses
Key Players: Moderna (U.S.), Pfizer-BioNTech (Germany/ U.S.), Alnylam Pharmaceuticals (U.S.), Ionis Pharmaceuticals (U.S.), CureVac (Germany), Arcturus Therapeutics (U.S.), Translate Bio (U.S.), Sarepta Therapeutics (U.S.), Sanofi (France), GSK (UK), Novartis (Switzerland), Regeneron (U.S.), Dynavax (U.S.), CSL Seqirus (Australia), GC Pharma (South Korea), Takeda Pharmaceutical (Japan), Daiichi Sankyo (Japan), Shionogi & Co. (Japan), Fujifilm Holdings (Japan), Mitsubishi Tanabe Pharma (Japan).
Global RNA Based Therapeutics Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 8.4 billion
- 2026 Market Size: USD 9.5 billion
- Projected Market Size: USD 26.2 billion by 2035
- Growth Forecasts: 13.5% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (42.3% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, China, Germany, Japan, United Kingdom
- Emerging Countries: India, South Korea, Singapore, Brazil, Israel
Last updated on : 28 August, 2025
RNA Based Therapeutics Market - Growth Drivers and Challenges
Growth Drivers
-
Rise in chronic diseases: Rare genetic conditions and neurodegenerative diseases comprise restricted treatment options that require advanced approaches, which denotes a huge opportunity for the RNA based therapeutics sector to flourish. According to the October 2024 CDC report, 10 people in America suffer from a chronic disorder, and 4 in 10 are affected by more than one chronic condition. Besides, the January 2024 NLM article reported that the approximate expense for chronic disease is USD 47 trillion, therefore denoting a huge opportunity for the market’s growth.
-
Surge in research and investment: Augmented funding and investments, breakthroughs in drug delivery, the availability of mRNA vaccines, and the presence of increased RNA therapeutics are effectively bolstering the RNA based therapeutics industry internationally. As stated in the February 2022 NLM report, 90% of clinical-specific drug development readily fails, despite multiple strategies, for which thorough research and generous investment are extremely essential. Besides, drug optimization has emphasized potencies due to which R&D activities are compulsory for bolstering the overall market.
-
Technological innovation: Advancements, including innovation in RNA and molecular platforms, optimized RNA synthesis and manufacturing, along with progressive delivery systems, are suitable for uplifting the RNA based therapeutics market. As stated in the February 2022 NLM report, the utilization of a synthetic oligonucleotide has been suitable for drug development and design, particularly in the pharmaceutical sector, and has successfully cleared 14 RNA-targeting drugs (RTDs). Therefore, with such an upliftment, the market is poised to grow in the upcoming years.
Global Disease Driving the Market
Prevalence Rate of Different Diseases
|
Diseases |
Incidences |
Year |
|
Cancer |
2,041,910 latest cases and 618,120 deaths |
2025 |
|
Duchenne Muscular Dystrophy |
1 in 3,600 males |
2023 |
|
Amyotrophic Lateral Sclerosis (ALS) |
0.2 per 100,000 person-years to 23.4 per 100,000 person-years |
2023 |
|
Huntington’s Disease |
0.3 per 100,000 person‐years to 2.7 per 100,000 persons |
2022 |
|
Familial Hypercholesterolemia Syndrome |
3,000 to 5,000 patients |
2023 |
|
Hereditary Transthyretin-mediated Amyloidosis (hATTR) |
5,000 to 38,000 individuals |
2023 |
|
Kidney Diseases |
>10% of the general population, catering to >800 million individuals |
2022 |
Sources: NIH, NLM
Cardiovascular Disease-Based Risks Boosting the Market
Risk Factors Associated with CVD Leading to Deaths (2023)
|
CVD-Based Risks |
Deaths |
|
Elevated LDL cholesterol |
3.8 million |
|
High fasting plasma glucose |
2.3 million |
|
Air pollution |
4.8 million |
|
High body-mass index |
2.0 million |
|
Tobacco use |
3.0 million |
|
Low physical activity |
397,000 |
|
Raised blood pressure |
10.8 million |
Sources: World Heart Federation Organization
Challenges
-
Fragmentation in regulation: The market is consistently witnessing interrupted clearance pathways across every nation, which is readily delaying market entry. For instance, the centralized process of the EMA usually takes months for approval, which has negatively impacted the overall market. Owing to this barrier, organizations have faced delays, owing to the demand for additional safety requirements. Besides, small-scale markets frequently wait for years to receive approval, which has further created disparities in accessibility, thus hampering the overall market growth.
-
Storage and cold chain expenses: The majority of RNA therapies need ultra-cold storage facilities, and in low-resource settings, there is a demand for adequate freezers, which has restricted the distribution, thereby negatively impacting the RNA based therapeutics sector. Companies’ vaccines usually need cold storage benefits, but rural regions are continuing to witness spoilage rates, owing to power outages. However, to combat this, organizations have created thermostable LNPs that can easily survive cold temperatures and reduce wastage in pilot programs.
RNA Based Therapeutics Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
13.5% |
|
Base Year Market Size (2025) |
USD 8.4 billion |
|
Forecast Year Market Size (2035) |
USD 26.2 billion |
|
Regional Scope |
|
RNA Based Therapeutics Market Segmentation:
End user Segment Analysis
Based on the end user, the biopharmaceutical companies segment in the RNA based therapeutics market is anticipated to garner the highest share of 58.6% by the end of 2035. The segment’s growth is augmented by an increase in drug development, the delivery of the latest therapies, and an increased adoption of artificial intelligence (AI). In this regard, the November 2024 ITIF report depicted that with drug development, only 8% early candidates are able to gain access. Besides, the R&D expense for launching a new drug to the market accounts for USD 2.8 billion, and capitalized at a yearly rate of 10.5%, which increases to USD 4.0 billion, thus suitable for the market to grow internationally.
Type Segment Analysis
Based on the type, the mRNA therapeutics segment in the RNA based therapeutics market is projected to account for the second-largest share of 48.5% during the forecast timeline. The segment’s upliftment is effectively propelled by the tactical focus on cutting-edge vaccine platforms, as well as cancer immunotherapies, suitable for mRNA infectious diseases. In addition, regional manufacturers, such as Takeda and Daiichi Sankyo, are readily investing to develop localized manufacturing facilities through collaboration with Tokyo-based contract developers. Meanwhile, the presence of a streamlined administrative pathway for mRNA products, which are approved by the PDMA, is also fueling the segment’s demand.
Application Segment Analysis
Based on the application, the oncology segment in the RNA based therapeutics market is expected to grab the third-largest share of 41.7% by the end of the projected period. The segment’s development is effectively driven by a surge in cancer cases, an increase in mortality, and advancements in resource settings. As per the January 2025 NLM article, there will be more than 24 million latest cancer cases, along with 13 million deaths internationally. In addition, approximately 17 million patients will require surgery for this condition, thereby accounting for 45 million operations by the end of 2030. Therefore, with all these possibilities, there is a huge opportunity for oncology to outshine in the market globally.
Our in-depth analysis of the market includes the following segments:
|
Segment |
Subsegments |
|
End user |
|
|
Type |
|
|
Application |
|
|
Delivery Mechanism |
|
|
Product |
|
|
Indication |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
RNA Based Therapeutics Market - Regional Analysis
North America Market Insights
North America in the RNA based therapeutics industry is anticipated to be the dominating region, garnering the highest share of 42.3% by the end of 2035. The market’s development in the region is highly attributed to continuous research and development (R&D), administrative benefits, an increase in healthcare expenditure, and robust partnerships. Besides, according to the 2025 World Bank Organization report, the current healthcare expenditure of the region in terms of gross domestic product (GDP) is 16.0%, which is also fueling the market. Meanwhile, the July 2024 NIH report stated that Gotham Therapeutics, which has acquired 858 therapeutics, along with Accent Therapeutics, raised a generous capital of USD 50 million each to create drugs for targeting RNA-based therapeutics, thus creating a boost in the market.
The market in the U.S. is continuously growing, owing to the provision of federal funding, reimbursement policies, and private sector leadership. In addition, clinical trial kits sourcing is yet another factor uplifting the market in the country. In this regard, the 2023 OEC report stated that the country is the top exporter of these kits, accounting for USD 294 million. Additionally, the import valuation is USD 144 million, thereby facilitating the administration and smooth delivery of RNS-based medications, particularly during clinical trials.
The RNA based therapeutics market in Canada is also growing significantly since there has been the initiation of government investments, provincial healthcare, and research ecosystem. According to the February 2024 Government of Canada report, there has been a generous contribution of USD 200,000 to RNA Technologies & Therapeutics Inc. to enable automated equipment for optimizing the overall production capacity. Moreover, small and medium enterprises (SMEs) in Quebec account for 99.7% of province-based businesses, along with 50% of the GDP, thereby suitable for bolstering the market’s growth in the country.
Past 14 Years Population Share Covered by Healthcare Insurance in North America (2024)
|
Countries in North America |
Population Share |
|
U.S. |
84% |
|
Mexico |
85.6% |
|
Canada |
100% |
|
Bahamas |
100% |
|
Jamaica |
20.1% |
Source: World in Data Organization
APAC Market Insights
Asia Pacific in the RNA based therapeutics market is expected to emerge as the fastest-growing region, grabbing a share of 22.4% during the forecast timeline. The market’s upliftment in the region is effectively fueled by government-based strategies, pharmaceutical investments, and administrative clearances. Besides, in April 2025, Marubeni Corporation made a share purchase agreement with Sumitomo Pharma Co., Ltd. to acquire its share in Asia, with a valuation of YEN 45 billion, and successfully take over by establishing a pharmaceutical strategic platform. Therefore, with such developments, the market is expected to boost in the overall region.
The market in China is gaining increased traction, owing to administrative initiatives, regional mRNA production, an increase in the patient demand, and export potentialities. According to the October 2024 NLM article, the dsRNA production rate among mRNA products was readily plotted at 100%. In addition, T7 RNAP G753A-catalyzed mRNA products diminished dsRNA impurities by 1.3%. Besides, as per the 2023 OEC data report, the country readily imports clinical trial kits, with a valuation of USD 74.9 million, thus creating an optimistic outlook for the overall market.
The RNA based therapeutics market in India is steadily progressing due to the presence of affordable manufacturing, private funding, a surge in the disease burden, and growth in clinical trials. In addition, organization collaboration is also another factor that is uplifting the market in the country. For instance, in March 2025, TechInvention Lifecare Pvt. Ltd. collaborated with Belgium-based Quantoom Biosciences, with the intention of making advancements in RNA-based vaccines. Based on this memorandum of understanding, TechInvention is projected to launch Quantoom’s next-generation RNA N-Force toolbox in the country, thereby denoting a huge growth opportunity for the market.
Europe Market Insights
Europe in the RNA based therapeutics market is projected to account for a considerable share of 28.1% during the forecast duration. The market’s development in the region is highly driven by administrative leadership, a robust R&D ecosystem, an increase in health and medical spending, and enhanced focus on vaccine and oncology. According to an article published by Vaccine in April 2023, 74.4% of the population in the overall region has access to vaccine since the COVID-19 onset. Therefore, this denotes the possibility of vaccine-based policy and reforms' presence, which denotes an optimistic outlook for the overall market.
The market in Germany is steadily growing, owing to BioNTech’s dominance, provision of funds from the government, manufacturing capacities, and increased focus on rare diseases. As per the May 2022 MSF Access Campaign report, BioNTech was provided with €500 million as a public fund from public investors, including €375 million from the German Research Ministry, along with a €100 million loan from the European Investment Bank. The purpose of this fund was to ensure continuous R&D activities for developing vaccines, thereby suitable for the overall market upliftment in the country.
The RNA based therapeutics market in the UK is also gaining increased traction, highly attributed to the NHS integration, genomics leadership, academic and industry partnership, and flexibility through Brexit. According to the June 2025 UK Government report, the UKHSA, along with NHS England, and DHSC are continuing to collaborate with each other, with NHS England leading to deliberately implement care systems in local areas. Besides, the 2023 OEC report stated that the country is one of the exporters of clinical trial kits, with a valuation of USD 107 million, thus effectively contributing towards the market’s growth.
Lifetime Vaccination Pricing in Europe
|
Components |
Price |
Median Range |
Diseases |
Median Values |
|
Healthy Individuals |
€592 to €3,504 |
€1,663 |
13 to 20 |
€1,203 |
|
Individuals with underlying conditions |
€744 to €9,081 |
€2,992 |
13 to 21 |
€1,731 |
|
Over scenario analysis |
€1,225 to €4,832 |
€1,731 |
21 to 22 |
€1,788 |
Source: NLM, January 2023
Key RNS Based Therapeutics Market Players:
- Moderna (U.S.)
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Pfizer-BioNTech (Germany/ U.S.)
- Alnylam Pharmaceuticals (U.S.)
- Ionis Pharmaceuticals (U.S.)
- CureVac (Germany)
- Arcturus Therapeutics (U.S.)
- Translate Bio (U.S.)
- Sarepta Therapeutics (U.S.)
- Sanofi (France)
- GSK (UK)
- Novartis (Switzerland)
- Regeneron (U.S.)
- Dynavax (U.S.)
- CSL Seqirus (Australia)
- GC Pharma (South Korea)
- Takeda Pharmaceutical (Japan)
- Daiichi Sankyo (Japan)
- Shionogi & Co. (Japan)
- Fujifilm Holdings (Japan)
- Mitsubishi Tanabe Pharma (Japan)
The international RNA based therapeutics market is severely competitive, with the presence of key players, including BioNTech, Pfizer, and Moderna, collectively dominating the global revenue. Besides, these companies have readily invested in cutting-edge mRNA platforms, as well as rare disease siRNA therapies. Meanwhile, Ionis and Alnylam are leading in RNAi, while Novartis and Sanofi successfully expanded into the oncology field. Furthermore, companies from Japan, such as Daiichi Sankyo and Takeda, have deliberately focused on localized mRNA production to diminish import dependency, thus suitable for the overall market’s development.
Here is a list of key players operating in the global market:
Recent Developments
- In January 2025, AstraZeneca declared an investment of CAD 820 million in Canada for developing over 700 job opportunities to conduct clinical studies, and redefine therapeutics for different diseases.
- In March 2024, DKSH entered into a distribution-based agreement with Southern RNA to successfully bolster its business opportunities and effectively develop its pharmaceutical business in India.
- Report ID: 8022
- Published Date: Aug 28, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
RNA Based Therapeutics Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




