Angioplasty Balloons Market Outlook:
Angioplasty Balloons Market size was valued at USD 3.6 billion in 2025 and is projected to reach USD 5.5 billion by the end of 2035, rising at a CAGR of 4.1% during the forecast period, i.e., 2026 to 2035. In 2026, the industry size of angioplasty balloons is assessed at USD 3.7 billion.

The continuously escalating patient pool affected by cardiovascular diseases is the key driver behind the steady growth of the market. This can be testified by the report from WHO published in July 2025 that states that nearly 19.8 million individuals lost their lives due to heart diseases in 2022, which marks 32% of overall worldwide deaths. The report also stated that out of those, 85% were due to heart attack and stroke. Therefore, the presence of this consumer base creates a heavy necessity for minimally invasive procedures such as angioplasty balloons.
Furthermore, the procedural costs and the payer dynamics, including insurance coverage, remarkably influence the market access and pricing strategies. Manufacturers are navigating this by demonstrating the long-term value of advanced products. In February 2025, the report from Beshak Organization revealed that angioplasty costs in developing nations such as India range from ₹80,000 to ₹2,00,000 i.e., USD 960 to USD 2,410, wherein certain insurers offering plans for individuals with pre-existing cardiac conditions, hence positively impacting market growth.
Key Angioplasty Balloons Market Insights Summary:
Regional Highlights:
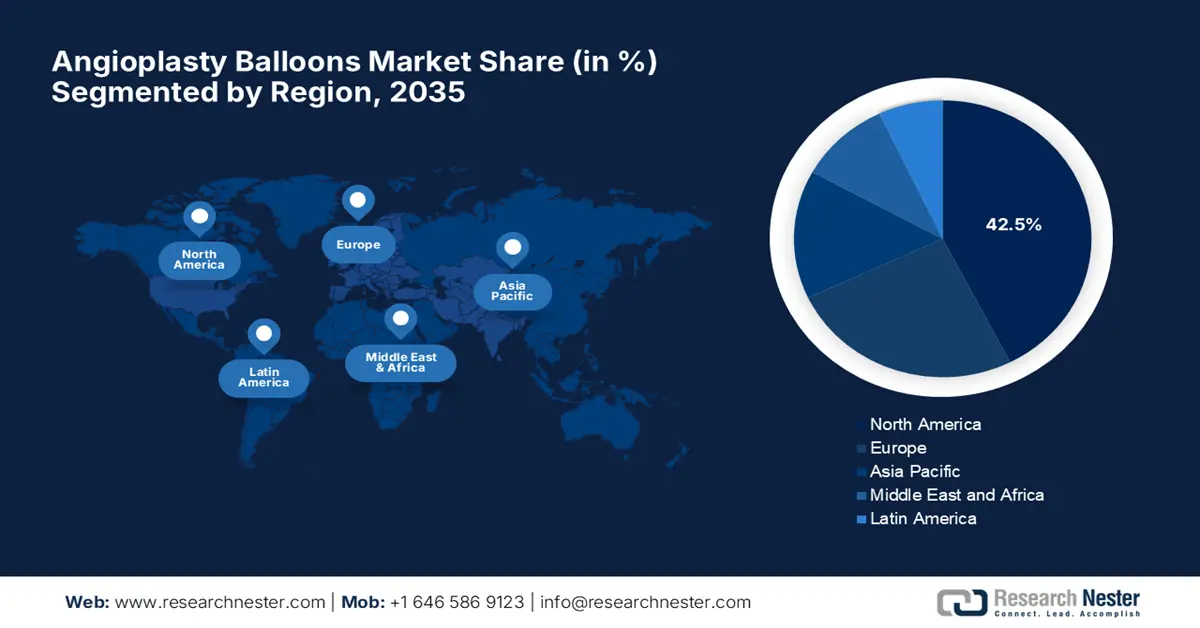
- North America is anticipated to command a 42.5% share by 2035 in the angioplasty balloons market, attributable to its advanced healthcare infrastructure and strong uptake of minimally invasive procedures.
- Asia Pacific is projected to witness rapid momentum by 2035 with its accelerating regional share, stemming from rising patient awareness and expanding healthcare infrastructure.
Segment Insights:
- The hospitals & ASCs segment is expected to secure a 78.6% revenue share during the forecast tenure in the angioplasty balloons market, bolstered by the availability of advanced labs and skilled personnel for percutaneous coronary interventions.
- The application segment is on track to capture a 65.4% share by 2035, supported by the increasing prevalence of coronary diseases associated with obesity, diabetes, and hypertension.
Key Growth Trends:
- Increasing awareness of early diagnosis
- Advancements in balloon catheter technology
Major Challenges:
- Amplifying procedure and device costs
- Increased risk of complications
Key Players: Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, B. Braun Melsungen AG, Cardinal Health (Cordis), BIOTRONIK SE & Co. KG, Koninklijke Philips N.V., BD (Becton, Dickinson and Company), Cook Medical LLC, QT Vascular Ltd., Meril Life Sciences Pvt. Ltd., Lepu Medical Technology (Beijing) Co., Ltd., MicroPort Scientific Corporation, OrbusNeich Medical, Endocor GmbH.
Global Angioplasty Balloons Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 3.6 billion
- 2026 Market Size: USD 3.7 billion
- Projected Market Size: USD 5.5 billion by 2035
- Growth Forecasts: 4.1% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (42.5% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, China, Germany, Japan, United Kingdom
- Emerging Countries: India, Brazil, South Korea, Mexico, Indonesia
Last updated on : 29 August, 2025
Angioplasty Balloons Market - Growth Drivers and Challenges
Growth Drivers
-
Increasing awareness of early diagnosis: The awareness programs backed by public and private entities play a crucial role in prompting adoption in the angioplasty balloons sector. For instance, in June 2025, Medanta initiated the Mission Save Heart campaign in Guwahati. Besides, the initiative focused on increasing awareness around hypertension, sleep apnea, and their impact on cardiovascular health. The event included a Continuing Medical Education (CME) conference for cardiologists, highlighting advancements in treatment and prevention amid the growing cardiovascular disease.
-
Advancements in balloon catheter technology: Tech-based discoveries in drug-coated balloons, specialty balloons have remarkably fueled market growth across different nations. In July 2025, Teleflex Incorporated notified that it received approval from the U.S. Food and Drug Administration (FDA) 510(k) for the Ringer Perfusion Balloon Catheter (PBC), which marks the only available percutaneous transluminal coronary angioplasty perfusion balloon. Thus, such instances encourage more pioneers to capitalize on this field.
-
Increasing adoption of minimally invasive procedures: There has been consistent progress in the adoption of these procedures since they enable a faster recovery and reduced hospital stays. In this regard, Medtronic in February 2025 stated that it successfully enrolled the first patient in its trial of the Prevail paclitaxel-coated balloon catheter, which is also known as a drug-coated balloon for in-stent restenosis (ISR) and de novo small vessel disease, hence indicating a positive market outlook.
Historic Regional Age-Standardized Cardiovascular Disease (CVD) Death Rates (per 100,000, 2019)
|
Region |
Female Death Rate |
Male Death Rate |
|
Central Europe, Eastern Europe, Central Asia |
345.7 |
524.1 |
|
North Africa & Middle East |
339.8 |
376.7 |
|
South Asia |
264.2 |
360.4 |
|
Sub-Saharan Africa |
287.7 |
315.9 |
|
Southeast Asia, East Asia, Oceania |
179.8 |
262.8 |
|
Latin America & Caribbean |
141.3 |
200.7 |
|
High-Income |
102.1 |
153.8 |
Source: World Heart Federation 2023
Cost of CHD by World Region 2022 (Subgroup Meta-Analysis)
|
Region |
Mean Annual Cost per Patient (% of GDP per capita) |
95% Confidence Interval |
|
North America |
47.74% |
32.35% to 63.14% |
|
Latin America & Caribbean |
17.39% |
-6.67% to 41.45% |
|
Europe & Central Asia |
16.47% |
8.41% to 24.54% |
|
East Asia & Pacific |
15.03% |
2.17% to 27.90% |
|
Middle East |
10.97% |
-32.15% to 54. |
Source: NIH January 2025
Challenges
-
Amplifying procedure and device costs: Despite the clinical evidence, the market still faces barriers with the comprehensive pricing ranges of these procedures, especially in developing nations. These high prices have skewed up the accessibility and affordability, restricting their adoption in this field. The existence of this economic challenge can ultimately slow down the market growth as both service providers and consumers seek cost-effective alternatives.
- Increased risk of complications: Manufacturers and investors involved in the angioplasty balloons industry often hesitate to operate in this field owing to the risk of complications such as restenosis, where the treated artery narrows again after the procedure. This, in turn, creates repeated interventions, thereby reducing success rates and patient confidence as well. Also, such complications push clinicians to weigh the benefits against potential drawbacks, thereby limiting the widespread adoption.
Angioplasty Balloons Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
4.1% |
|
Base Year Market Size (2025) |
USD 3.6 billion |
|
Forecast Year Market Size (2035) |
USD 5.5 billion |
|
Regional Scope |
|
Angioplasty Balloons Market Segmentation:
End user Segment Analysis
Based on end user hospitals & ASCs segment is projected to gain the largest revenue share of 78.6% in the market during the forecast tenure. These primary facilities are equipped with advanced labs and a skilled workforce for performing percutaneous coronary interventions positioning the segment at the forefront to generate revenue in the sector. In November 2021, Robocath reported that it successfully completed its first robotic-assisted coronary angioplasty in China using the R-One platform at Beijing’s 301, thus reflecting a significant milestone in the growing adoption of innovative technologies in cardiovascular treatments.
Application Segment Analysis
In terms of the application segment is expected to capture a share of 65.4% in the angioplasty balloons market by the end of 2035. The growing burden of coronary diseases influenced by a rise in obesity, diabetes, and hypertension is the key factor behind the leadership. In March 2025, Abbott notified that the U.S. FDA had accepted an investigational device exemption for its Coronary Intravascular Lithotripsy (IVL) System to analyze treatment severity in severe calcification coronary arteries prior to stenting, hence denoting a positive segment outlook.
Product Type Segment Analysis
Based on product type drug-eluting balloons segment is expected to grow at a considerable rate, with a share of 42.7% in the angioplasty balloons market during the discussed timeframe. The growth in the segment is subject to their superior efficacy in treating stent restenosis and small vessel diseases. In March 2024, Boston Scientific received FDA approval for the AGENT Drug-Coated Balloon, the first coronary DCB approved in the U.S. for treating coronary in-stent restenosis (ISR). The company also stated that the device offers a safe and effective alternative to address vessel narrowing caused by plaque or scar tissue after stent placement.
Our in-depth analysis of the market includes the following segments:
|
Segment |
Subsegments |
|
End user |
|
|
Application |
|
|
Product Type |
|
|
Ball0on Type |
|
|
Disease Indication |
|
|
Material |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Angioplasty Balloons Market - Regional Analysis
North America Market Insights
North America is considered to be the dominating region in the global angioplasty balloons market, holding the largest share of 42.5% by the end of 2035. The region’s advanced healthcare infrastructure and strong adoption of minimally invasive procedures are the key factors behind this proprietorship. In this regard, Boston Scientific in October 2023 reported the positive 12-month results from the pivotal AGENT IDE clinical trial, demonstrating the superiority of the AGENT Drug-Coated Balloon (DCB) over uncoated balloon angioplasty in treating coronary in-stent restenosis (ISR). Such instances highlight a major potential for the market to progress in this region.
The U.S. is augmenting its leadership in the regional angioplasty balloons market with the enlarging patient pool of coronary artery disease and peripheral vascular disorders. Also, the presence of international leaders allows continuous introduction of novel balloon catheter designs, enabling enhanced patient outcomes. In March 2024, Beth Israel Deaconess Medical Center (BIDMC) declared the results from the largest randomized clinical trial in the U.S., demonstrating that drug-coated balloons coated with paclitaxel appreciably reduce restenosis rates in patients undergoing coronary angioplasty when compared to uncoated balloons.
Canada in the angioplasty balloons market is gaining enhanced recognition, effectively attributed to the substantial funding grants and increasing awareness of cardiovascular health. Continued advancements in medical technology and the presence of leading device manufacturers also foster a competitive landscape. In May 2022, the Government of Canada, through the Canadian Institutes of Health Research (CIHR), announced a total investment of USD 5 million to establish the Canadian Heart Function Alliance, a national research network focused on heart failure, hence suitable for standard market development.
U.S. Cardiovascular Disease: Key Figures
|
Statistic |
Value |
|
Death frequency |
1 death every 34 seconds |
|
Total deaths from CVDs (2023) |
919,032 deaths |
|
Proportion of total deaths (2023) |
1 in every 3 deaths |
|
Economic cost (2020 - 2021) |
$417.9 billion |
|
Deaths from coronary artery disease (2022) |
371,506 deaths |
|
Annual heart attacks (total) |
805,000 events |
Source: CDC
APAC Market Insights
Asia Pacific is expected to be the fastest-growing region in the global angioplasty balloons market by the end of 2035. This rapid pace of progression originates from the rising patient awareness and the expanding healthcare infrastructure. Besides the vigorous escalation of technological discoveries and government initiatives to improve cardiac care, market adoption is also readily accelerating market adoption. In addition, the region hosts encouraging reimbursement frameworks that create strong opportunities for innovative balloon catheter technologies.
China is maintaining a leading position in the angioplasty balloons sector on account of a strong government focus on improving cardiovascular health, coupled with increased investments in healthcare infrastructure and research. For instance, in February 2025, BrosMed and Cordis entered into a strategic alliance to distribute BrosMed’s next-generation drug-coated peripheral balloon catheter, VaSecure, in the country’s market. This collaboration is expected to advance peripheral angioplasty balloon technology, thereby strengthening the country’s position in this field.
India in the angioplasty balloons market is growing steadily, effectively attributed to the cardiovascular disease burden and expanding access to healthcare services. The country’s government is constantly putting efforts into improving cardiac care infrastructure, which also fosters a favorable business environment. In this context, in March 2023, Kauvery Hospital successfully performed a coronary angioplasty using drug-coated balloons on a 74-year-old patient, by avoiding the use of metal stents. Besides, this image-guided procedure preserved artery function and enabled a promising alternative to traditional stenting.
Angiography Cost in Asia-Pacific Countries 2024
|
Country |
Cost (USD) |
|
India |
$300 |
|
Thailand |
$4,200 |
|
Singapore |
$13,400 |
|
Malaysia |
$8,000 |
|
South Korea |
$17,700 |
Source: AHI
Europe Market Insights
Europe is portraying steady growth in the worldwide market of angioplasty balloons due to the presence of rapidly aging demographics. The strong regulatory frameworks and increased adoption of minimally invasive procedures contribute to market expansion. Exemplifying such a supportive atmosphere EU in October 2023 reported that transluminal coronary angioplasty procedures rose significantly across most European countries, reflecting their preference over bypass surgeries for treating cardiovascular diseases. The study also revealed that over 1.1 million angioplasties were performed in 25 member states. Croatia reported the highest procedure rate, followed by Germany and Latvia.
Germany is augmenting its leadership in the regional angioplasty balloons market with widespread access to cutting-edge technologies and a strong emphasis on minimally invasive cardiac interventions. In May 2025, Demax Group reported that it had established its wholly owned subsidiary, called Demax Medical Germany GmbH, which focuses on coronary, neuro, and peripheral interventions, with a strong presence in over 90 countries and a significant market share in coronary artery accessories, hence suitable for standard market development.
The U.K. plays a pivotal role in solidifying the region’s progress in the market, empowered by the national health initiatives focused on reducing cardiovascular disease burden. Continuous investments in healthcare technology and the rising demand for advanced angioplasty balloons and drug-eluting devices across the country also provide encouraging opportunities for players to capitalize on this field. In September 2024, the Royal London Hospital became the first in Europe to implement the innovative EMBOGUARD balloon guide catheter in clinical practice for mechanical thrombectomy, a crucial stroke treatment.
Key Angioplasty Balloons Market Players:
- Medtronic plc
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Boston Scientific Corporation
- Abbott Laboratories
- B. Braun Melsungen AG
- Cardinal Health (Cordis)
- BIOTRONIK SE & Co. KG
- Koninklijke Philips N.V.
- BD (Becton, Dickinson and Company)
- Cook Medical LLC
- QT Vascular Ltd.
- Meril Life Sciences Pvt. Ltd.
- Lepu Medical Technology (Beijing) Co., Ltd.
- MicroPort Scientific Corporation
- OrbusNeich Medical
- Endocor GmbH
The global market is extremely consolidated, which is dominated by the large pioneers such as Medtronic, Boston Scientific, and Abbott. The players are readily investing in R&D to develop next-generation drug-eluting balloons and specialty balloons due to their enhanced safety profiles and consumer preferences. Also, mergers & acquisitions, geographic penetration are expanding product portfolios, thereby fostering a greater business environment. Furthermore, regulatory acceptances for wider applications are also solidifying the firm’s market positions.
Below is the list of some prominent players operating in the global market:
Recent Developments
- In January 2025, Bentley announced that it had launched two new stent graft systems, called BeGraft Stent Graft System and BeFlared FEVAR Stent Graft System is the first dedicated bridging stent indicated specifically for FEVAR procedures.
- In January 2025, Concept Medical Inc. announced it successfully finished the first patient enrolment in the MAGICAL BTK, which is a randomized controlled trial of MagicTouch PTA. This is for Sirolimus Coated Balloon Versus Standard Balloon Angioplasty in the treatment of below-the-knee arterial disease.
- Report ID: 8030
- Published Date: Aug 29, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Angioplasty Balloons Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




