Rapid Antigen Testing Market Outlook:
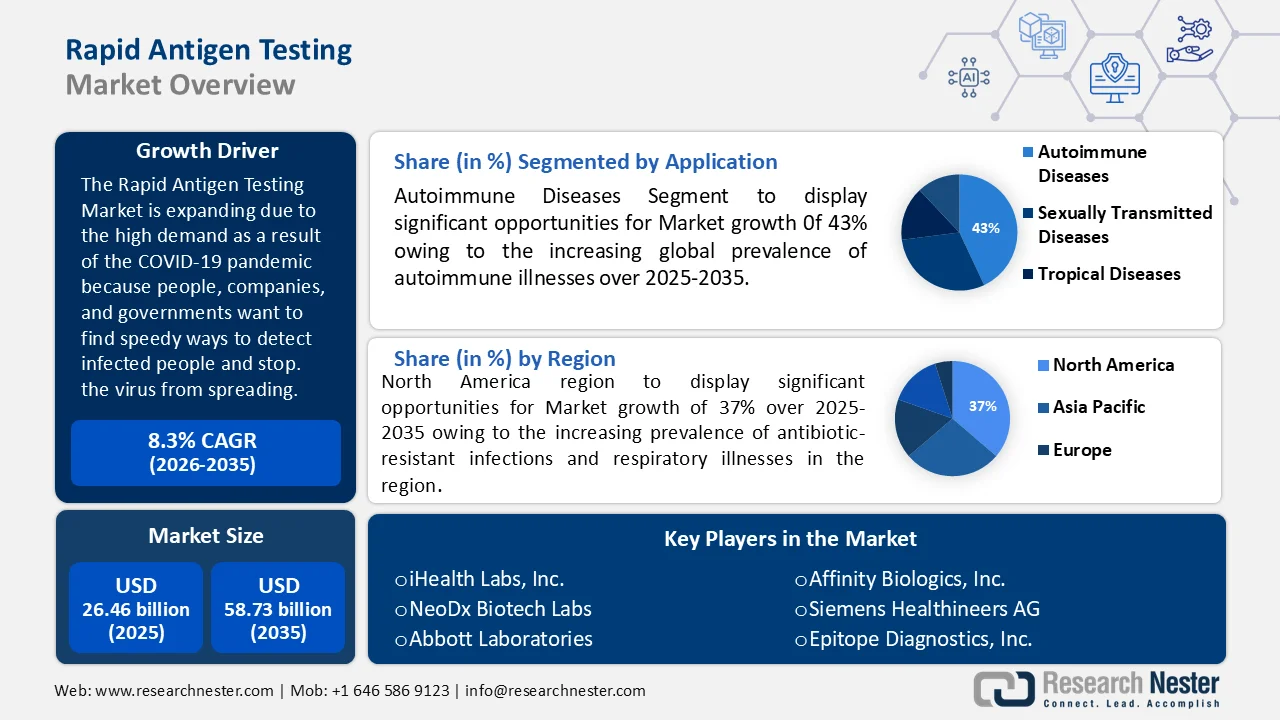
Rapid Antigen Testing Market size was over USD 26.46 billion in 2025 and is projected to reach USD 58.73 billion by 2035, witnessing around 8.3% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of rapid antigen testing is evaluated at USD 28.44 billion.
Rapid antigen testing is in high demand as a result of the COVID-19 pandemic because people, companies, and governments want to find speedy ways to detect infected people and stop the virus from spreading.
Also, the accuracy provided by these tests during the COVID-19 increased their demand during the pandemic. According to a study in 2022, antigen testing accurately detected COVID-19 infection in 73% of symptomatic individuals with confirmed COVID-19, compared to 55% of asymptomatic individuals. Rapid antigen testing is a crucial weapon in the pandemic response since it can yield results in as little as 15 minutes.
Several regulatory bodies, including the US FDA, the European Union, and other national regulatory bodies, have approved rapid antigen testing for use which will drive the rapid antigen testing market demand to increase rapidly. Rapid antigen testing is now more widely available and used as a result of this. For instance, the Flowflex COVID-19 Antigen Home Test, the first and second COVID-19 at-home tests to satisfactorily pass traditional premarket evaluation, was approved for commercialization by the Food and Drug Administration on November 9.
Key Rapid Antigen Testing Market Insights Summary:
Regional Insights:
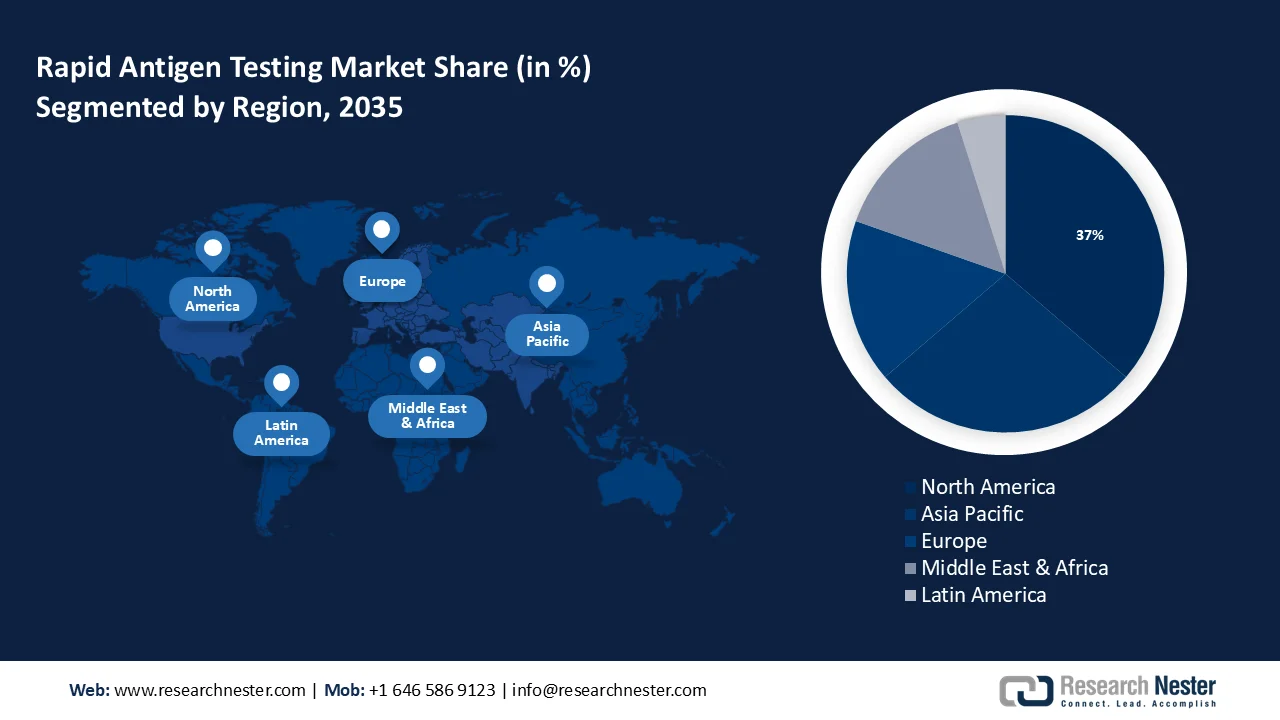
- North America is projected to command a 37% revenue share of the rapid antigen testing market by 2035, attributed to the rising prevalence of antibiotic-resistant infections and respiratory illnesses.
- Asia Pacific is expected to capture over 28% revenue share by 2035, propelled by the growing number of infectious disease cases and increasing healthcare investments across the region.
Segment Insights:
- In the rapid antigen testing market, the autoimmune diseases segment is poised to hold more than 43% share by 2035, driven by the increasing global prevalence of autoimmune illnesses.
- The immunosorbent assay segment is estimated to account for over 33% revenue share by 2035, fueled by its extensive application in detecting antigens in cerebrospinal fluid for meningitis-causing organisms.
Key Growth Trends:
- Growing adoption of rapid antigen testing kits for diagnosis of other viral infections
- Growing shift towards digitalization in the healthcare industry
Major Challenges:
- False test results may impede the market growth
- Stringent regulations by the government for the approval of rapid testing products.
Key Players: Dignity Health, Emerus Hospital Partners, LLC, SCL Health, Saint Luke's Health System, Baylor Scott & White Health, CHRISTUS Health, Amerisource Bergen Corp., Cardinal Health Inc., Cigna Corp., Elevance Corp., Pfizer, Walgrees Boots Alliance Inc., Trivitron Healthcare, Seaskymedical.
Global Rapid Antigen Testing Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 26.46 billion
- 2026 Market Size: USD 28.44 billion
- Projected Market Size: USD 58.73 billion by 2035
- Growth Forecasts: 8.3% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (37% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, China, United Kingdom, Japan
- Emerging Countries: China, India, Brazil, Germany, Japan
Last updated on : 25 February, 2026
Rapid Antigen Testing Market - Growth Drivers and Challenges
Growth Drivers
- Growing adoption of rapid antigen testing kits for diagnosis of other viral infections - For the diagnosis of RSV or influenza, among other viral infections, rapid antigen testing may be helpful. The rapid antigen testing market for these tests may grow if businesses take the opportunity to create quick antigen tests for these additional viral illnesses.
- Growing shift towards digitalization in the healthcare industry - The advancement of technology, particularly the internet and mobile devices, has brought about an enormous transformation in the healthcare industry. Tools like electronic health records, online appointments, and safe data storage have been utilized by both patients and healthcare professionals at healthcare institutions. Rapid antigen testing can be combined with digital technology, including cloud-based platforms or smartphone apps, to expedite the reporting and testing procedures.
For instance, a leading global provider of medical technology, BD (Becton, Dickinson, and Company), announced the immediate availability of the ImageMover app in Canada. This mobile app is a companion to the BD VeritorTM Plus System and allows for more efficient management of SARS-CoV-2 antigen testing results, thereby lowering the need for manual documentation. - Growing incidences of infectious diseases - Rapid and effective diagnostic methods are becoming more and more necessary due to the rising prevalence of infectious disorders like strep throat, influenza, and respiratory infections. As per a report, the average annual death toll from respiratory diseases is over 400,000.
When new strains have emerged in big pandemics, the mortality toll has been significantly higher. Quick antigen testing provides results in a matter of minutes, facilitating prompt identification and suitable treatment of the illness. This facilitates prompt medical attention and helps stop the illness from spreading to other people. Therefore, the growing incidences of infectious diseases have increased the demand for rapid influenza diagnostics, which is propelling the growth of the rapid antigen testing market
Challenges
- False test results may impede the market growth - Rapid antigen testing does have certain restrictions, though. They have a higher percentage of false negative results and are less accurate than NAAT/PCR testing, especially when it comes to identifying asymptomatic cases.
Therefore, if there is a strong suspicion of COVID-19 infection, negative results from fast antigen tests should be verified by an NAAT/PCR test. - Limited awareness regarding advanced rapid antigen testing which may restrain the rapid antigen testing market from growing.
- Stringent regulations by the government for the approval of rapid testing products.
Rapid Antigen Testing Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
8.3% |
|
Base Year Market Size (2025) |
USD 26.46 billion |
|
Forecast Year Market Size (2035) |
USD 58.73 billion |
|
Regional Scope |
|
Rapid Antigen Testing Market Segmentation:
Application Segment Analysis
In rapid antigen testing market, autoimmune diseases segment is poised to hold more than 43% share by 2035. The advancement of the segment is significantly driven by the increasing global prevalence of autoimmune illnesses. The annual increases in the incidence and prevalence of autoimmune illnesses globally are estimated to be 19.1% and 12.5%, respectively.
Millions of people throughout the world suffer from autoimmune illnesses like psoriasis, lupus, rheumatoid arthritis, and multiple sclerosis. Also, early identification and diagnosis of autoimmune disorders have been made easier by developments in diagnostic technology. Healthcare professionals may now diagnose autoimmune illnesses early on, improving patient outcomes and starting therapy sooner because of the development of more precise and dependable diagnostic techniques.
Research and development related to autoimmune diseases are advancing quickly. As new understandings of the fundamental processes underlying autoimmune disorders emerge, prospective therapeutic targets and methods of care are being identified.
Technique Segment Analysis
In rapid antigen testing market, immunosorbent assay segment is estimated to account for more than 33% revenue share by the end of 2035. The main application of immunosorbent assay is the detection of antigens in the cerebrospinal fluid (CSF) of meningitis patients' Hemophilus, Streptococcus, and Cryptococcus organisms.
These are widely employed in the diagnosis of Lyme disease, HIV, and viral hepatitis. Therefore, the growing cases of these diseases will propel the growth of the segment. According to a report, more than 1.34 million people worldwide lose their lives to viral hepatitis each year, a mortality toll that is equivalent to that of AIDS and tuberculosis. In addition, globally, chronic hepatitis is the cause of 78% of cases of liver cancer.
Moreover, Directigen, QuickVue, Sofia, and BD Veritor are next-generation lateral flow Digital Immunoassay (DIA) techniques for the qualitative identification of RSV viral antigens. These are immunoassay-based fast diagnostics for RSV. Rapid RSV antigen testing is also the most popular test. Most RSV antigen tests are done at an emergency room, doctor's office, or on-site, and most results are available in an hour or less.
Our in-depth analysis of the global rapid antigen testing market includes the following segments:
|
Technique |
|
|
Application |
|
|
End-User |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Rapid Antigen Testing Market - Regional Analysis
North American Market Insights
North America industry is anticipated to hold largest revenue share of 37% by 2035, rapid antigen testing market share due to the increasing prevalence of antibiotic-resistant infections and respiratory illnesses, the market is growing. According to a conservative estimate from the US Centers for Disease Control and Prevention (CDC), at least 23,000 Americans lose their lives to antibiotic-resistant diseases each year, impacting over two million individuals annually.
Additionally, the regional governments have put laws and programs into place to encourage the adoption of fast antigen testing. This entails supplying funds, increasing the number of tests available, and utilizing these tests in public health plans. Furthermore, the rapid antigen testing market for rapid antigen testing is expanding more quickly due to the existence of significant players like Abbott Laboratories, Quidel Corporation, and Roche Diagnostics.
APAC Market Insights
By 2035, Asia Pacific region in rapid antigen testing market is anticipated to capture over 28% revenue share due to rise in the number of infectious disease cases in the region is the reason for the growth. Additionally, governments are making significant investments in the healthcare sector, along with the people in the region are becoming more conscious of the necessity of quick antigen testing.
Furthermore, Asia-Pacific presents a profitable opportunity for businesses in the rapid testing sector because of its sizable population base and increasing level of disposable income. According to a report, the Asia and Pacific region is home to 60% of the world's population (4.3 billion people), including the world's most populated countries, China and India.
Rapid Antigen Testing Market Players:
- iHealth Labs, Inc.
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- NeoDx Biotech Labs
- Abbott Laboratories
- Affinity Biologics, Inc.
- Siemens Healthineers AG
- Epitope Diagnostics, Inc.
- Sino Biological, Inc.
- R-Biopharma AG
- Cell Biolabs, Inc.
- Thermo Fisher Scientific
Recent Developments
- iHealth Labs, Inc., a digital health startup and the United States' leading provider of at-home COVID tests, launched its Home Collection Kit for syphilis and HIV testing. Anyone over the age of 18 can use the FDA-approved method to take a blood sample in the privacy of their own home to test for syphilis and HIV (I, II, P24 antigen). They can mail the sample to the lab with a pre-paid overnight label and obtain accurate, private results in as little as two days.
- NeoDx Biotech Labs, a Bengaluru-based business, has introduced an in vitro diagnostic (IVD) kit for Ankylosing Spondylitis that uses real-time PCR technology. This kit enables healthcare services to enhance their testing capabilities. The HLA-B27 RT-PCR detection kit for Exon 2/Exon 3 is a real-time PCR test kit that is used to identify Human Leukocyte Antigen B27 in whole blood. HLA-B27 has a strong correlation with the inflammatory conditions of Ankylosing Spondylitis (AS), Psoriasis, Inflammatory Bowel Disease, and Reactive Arthritis. HLA-B27 testing can help in the early diagnosis and treatment of various conditions.
- Report ID: 5849
- Published Date: Feb 25, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Rapid Antigen Testing Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




