Essential Thrombocythemia Market Outlook:
Essential Thrombocythemia Market size was over USD 965.81 million in 2025 and is poised to exceed USD 1.73 billion by 2035, growing at over 6% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of essential thrombocythemia is estimated at USD 1.02 billion.
The market growth is primarily owing to rising cases of essential thrombocythemia (ET). Essential thrombocythemia impacts 1 to 24 individuals among every 1 million people, worldwide. This condition is characterized by the overproduction of platelets in the bone marrow, leading to an increased risk of blood clots. As more people are diagnosed with ET, the demand for effective treatment options and therapies to manage the condition increases, thereby driving the market.
Besides this, special programs by regulatory bodies to facilitate drug development will be an essential factor for essential thrombocythemia market growth. The FDA is putting together a set of four approaches to patient-focused drug development (PFDD) guidelines to aid in the advancement and implementation of organized methods for gathering and utilizing reliable and meaningful patient and opinions of caretakers that can facilitate better drug development and regulatory decision-making. Special programs like these assess the safety and efficacy of new treatments through rigorous clinical trials. Once approved, these drugs can be marketed and made available to patients, driving the ET market by expanding treatment options.
Key Essential Thrombocythemia Market Insights Summary:
Regional Highlights:
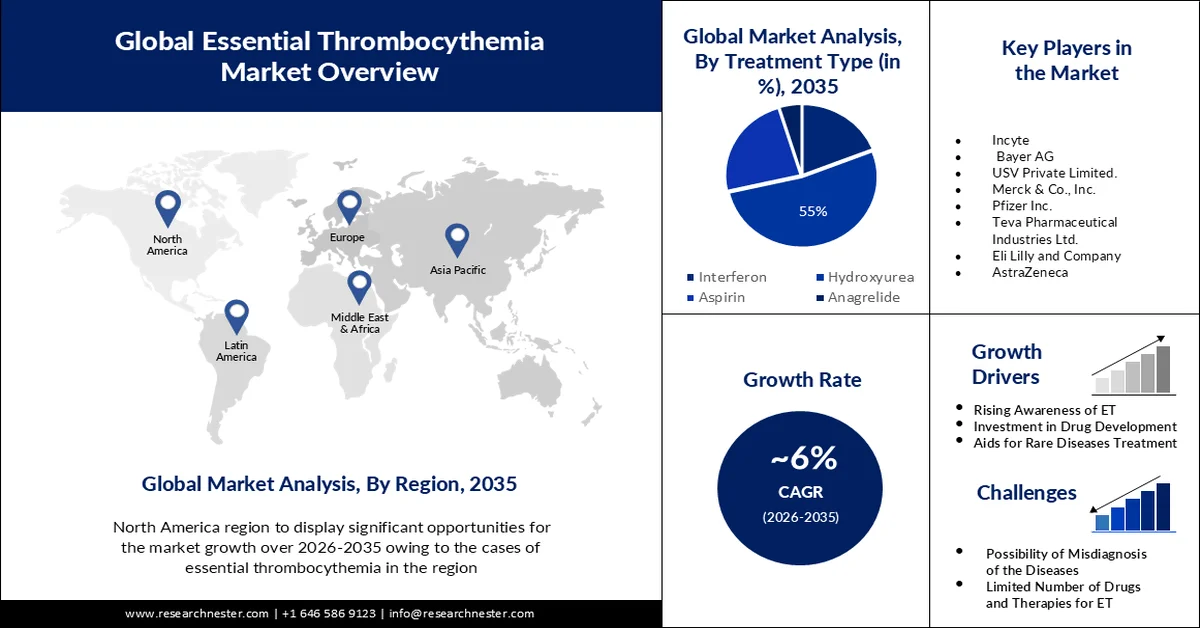
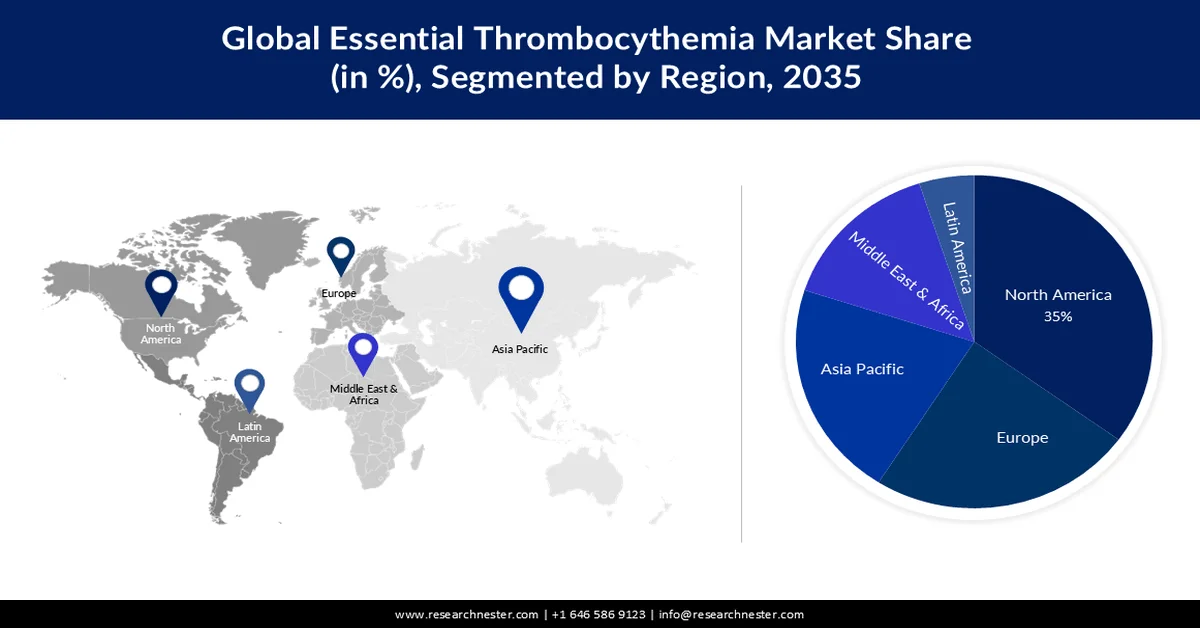
- North America is projected to hold a 35% share of the essential thrombocythemia market by 2035, propelled by the rising prevalence of ET and significant investments in the biotech and pharmaceutical sectors.
- Europe is expected to account for approximately 24% share by 2035, supported by increased health R&D investments fostering the development of personalized ET therapies.
Segment Insights:
- The hydroxyurea segment of the essential thrombocythemia market is projected to account for nearly 55% revenue share by 2035, stimulated by the rising cases of sickle cell anemia leading to secondary essential thrombocythemia.
- The research organization segment is forecast to capture 46% share by 2035, reinforced by advancements in genetic profiling, molecular diagnostics, and the identification of novel therapeutic targets.
Key Growth Trends:
- Government Initiatives for Rare Diseases
- Continuous Development of Treatment
Major Challenges:
- Lack of Opportunities for Clinical Trials
- Delayed diagnosis and treatment
Key Players: SkySpecs, Aerialtronics DV B.V., AeroVision Canada, AIRPIX, DJI, DroneView Technologies, HUVRdata, Intel Corporation, Microdrones GmbH, Nearthlab.
Global Essential Thrombocythemia Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 965.81 million
- 2026 Market Size: USD 1.02 billion
- Projected Market Size: USD 1.73 billion by 2035
- Growth Forecasts: 6% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (35% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, United Kingdom, France
- Emerging Countries: China, India, Brazil, South Korea, Mexico
Last updated on : 25 February, 2026
Essential Thrombocythemia Market - Growth Drivers and Challenges
Growth Drivers
-
Government Initiatives for Rare Diseases –The US National Institutes of Health and the European Commission, launched a joint commission in 2011, International Rare Disease Research Consortium (IRDiRC). Currently, the consortium has 60 organizations all across the world focusing on rare diseases. Moreover, IRDiRC has targeted new plans to reach nearly new therapies for rare diseases by the end of 2027. These initiatives can expedite the review and approval process for orphan drugs, which are drugs developed specifically for rare conditions like ET. Moreover, streamlined regulatory pathways enable faster access to innovative therapies, encouraging investment and development in the ET market.
-
Continuous Development of Treatment– The development of new and more effective treatments for ET leads to improved patient outcomes. Moreover, the availability of a diverse range of treatment options is crucial for addressing the varying need of ET patients. For instance, Bomedemstat Tosylate is in the second phase of development and it is primarily used for the treatment of essential thrombocythemia, and post-essential thrombocythemia myelofibrosis (PET-MF).
- Higher Prevalence in Old Adults – In the world, there were around 1 billion old adults in 2019. The population share is expected to rise and reach nearly 1.4 billion and 2.1 billion by 2030 and 2050, respectively. Aging is associated with an increased risk of developing essential thrombocythemia. As individuals age, the accumulation of genetic mutation and exposure to environmental factors over time may contribute to the development of ET. It is most commonly diagnosed in the sixth and seventh decade of an individual’s life.
Challenges
-
Lack of Opportunities for Clinical Trials – Conducting clinical trials for rare diseases like ET can be challenging due to the relatively small patient population and the need for specialized centers and expertise. Limited clinical trial opportunities can slow down the development of new therapies and hinder advancements in the field
-
Delayed diagnosis and treatment
- Limited Treatment Options
Essential Thrombocythemia Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
6% |
|
Base Year Market Size (2025) |
USD 965.81 million |
|
Forecast Year Market Size (2035) |
USD 1.73 billion |
|
Regional Scope |
|
Essential Thrombocythemia Market Segmentation:
Treatment Type (Interferon, Anagrelide, Hydroxyurea, Aspirin)
The hydroxyurea segment in the essential thrombocythemia market is anticipated to gain the largest revenue share of about 55% in the year 2035. The rising cases of sickle cell anemia will grow the segment’s growth. Hydroxyurea is considered a disease-modifying therapy for sickle cell anemia. Sickle cell anemia leads to secondary essential thrombocythemia. In sickle cell anemia, chronic inflammation and tissue damage associated with vaso-occulusive crises can stimulate the production of platelets. This can lead to elevated platelet levels in individuals. Every year, approximately 300,000 infants worldwide are born with a serious hemoglobin disorder. Moreover, roughly 250 million people worldwide are currently carriers of the gene responsible for sickle cell disease and associated hemoglobin disorders.
End User (Hospitals, Research Organizations, Specialty Clinics)
The research organization segment is expected to hold 46% share of the global essential thrombocythemia market in the year 2035. Moreover, the segment was responsible for around 65% of the market share in 2022. Researchers are leveraging innovative laboratory methods, genetic profiling, and molecular diagnostics to better understand the genetic and molecular basis of ET. These advancements not only aid in accurate diagnosis but also provide insights into disease progression and potential therapeutic targets. Research organizations are focused on identifying new therapeutic targets for essential thrombocythemia. By unraveling the underlying molecular and genetic abnormalities associated with ET.
Our in-depth analysis of the global market includes the following segments:
|
Treatment Type |
|
|
End User |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Essential Thrombocythemia Market - Regional Analysis
North America Statistics
North America industry is predicted to dominate majority revenue share of 35% by 2035. The market growth in the region is mostly driven by the rising count of people living with ET. In the United States, the average yearly prevalence of newly detected cases of ET for every ethnic group and race is roughly 2.2 per 100,000 population. Furthermore, the higher investment influx and development of biotech and pharmaceutical sectors are further driving the industry expansion in North America. Since, ET requires long-term management, often with the use of medication and these medications rely on the availability of high-quality active pharmaceutical ingredients (API), raw materials, and antibiotics. The Department of Health and Humans services invested USD 40 million to drive the growth of biopharmaceuticals in the United States.
European Market Forecast
The European essential thrombocythemia market is expected to be the second largest, with a share of around 24% by the end of 2035. According to the European Commission, by December 2021, European Union-based enterprises have raised their investments in health research and development by up to 10.3%. Increased investment in health R&D enables researchers to gain a deeper understanding of ET, including its underlying causes, mechanism, and risk factors. This knowledge contributes to the development of more personalized therapies for ET.
Essential Thrombocythemia Market Players:
- Active Biotech AB
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Incyte
- Bayer AG
- USV Private Limited.
- Merck & Co., Inc.
- Pfizer Inc.
- Teva Pharmaceutical Industries Ltd.
- Eli Lilly and Company
- AstraZeneca
- Takeda Pharmaceutical Company Limited
Recent Developments
- Active Biotech AB announced the approval for tasquinimod Orphan Drug Designation from the U.S. Food and Drug Administration (FDA) for the treatment of myelofibrosis. It creates an important regulatory channel and gives us the opportunity to rapidly progress tasquinimod development in this patient population.
- Incyte has announced to receive data from two Limber trials assessing single-agent therapy and combination treatments in patients with myelofibrosis (MF) using ruxolitinib with parsaclisib.
- Report ID: 5044
- Published Date: Feb 25, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Essential Thrombocythemia Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




