Biosimilars Market Outlook:
Biosimilars Market size was valued at USD 36.08 billion in 2025 and is expected to reach USD 161.93 billion by 2035, expanding at around 16.2% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of biosimilars is assessed at USD 41.34 billion.

The market growth can be ascribed to growing spending on biologics worldwide. Moreover, biologics are medications generated from live organisms that are used to treat a variety of ailments, including cancer, autoimmune disorders, and inflammations. They are often more expensive than ordinary small-molecule medications and can consume a sizable percentage of healthcare spending. It was found that, in Europe, biologics represented almost 34% of medicine spending at list prices, comprising small molecules, biologics, and biosimilar competitors.
Biosimilars help increase access to healthcare for many as it is responsible for the reduction in the healthcare costs associated with the use of biologics. Since biosimilar drugs are a very similar copies of their reference biologics, they require much less research and development for equal efficacy and safety than their reference biologics. The recent approvals of biosimilar drugs are set to reduce the spending on biologics, resulting in the demand for biosimilars among healthcare service providers, which in turn, is expected to create massive revenue generation opportunities for the global biosimilars market. It is estimated that biosimilars are expected to reduce direct spending on biologic drugs in the United States by USD 54 billion from 2017 to 2026, or around 3% of the total estimated biologic spending over the same period.
Key Biosimilars Market Insights Summary:
Regional Insights:
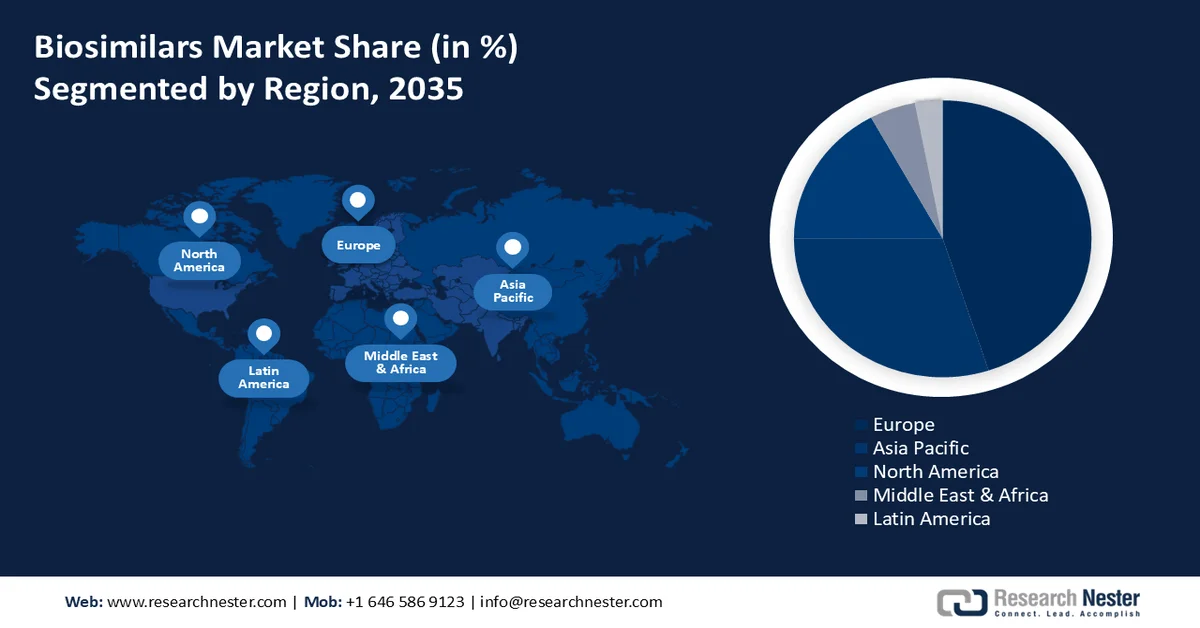
- Europe is projected to account for the largest revenue share of the biosimilars market by 2035, owing to the presence of a strong healthcare network, increasing number of hospitals and diseases, rising awareness among healthcare providers, and surge in biosimilar prescribing generating high savings from competition
- Asia Pacific is expected to witness notable expansion by 2035, attributed to improving healthcare infrastructure and growing adoption of cost-effective biologic alternatives
Segment Insights:
- The recombinant non-glycosylated proteins segment is poised to capture the highest share of the biosimilars market by 2035, impelled by the growing concern of rising incidences of diabetes along with the surge in the spread of diabetes worldwide
- The oncology segment will account for the largest share by 2035, driven by the rising number of cancer cases across the globe
Key Growth Trends:
- Upsurge in the Approval Rates of Biosimilars
- Growing Number of Cancer Cases
Major Challenges:
- Increasing Competition in the Market
- Manufacturing Process Concerns for Biosimilar Agents
Key Players: Weatherman, Upper Echelon Products LLC, FanHE Umbrellas, London Undercover Limited, Raintec Umbrellas, Shaoxing Shangyu Yuzi Outdoor Products Co., Ltd., Happy Umbrella Foundation, Citizen Watch Co., Ltd., Woodline Shade Solutions PTY Ltd, Uhlmann Sonnenschirme e.K., Fulton Umbrellas.
Global Biosimilars Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 36.08 billion
- 2026 Market Size: USD 41.34 billion
- Projected Market Size: USD 161.93 billion by 2035
- Growth Forecasts: 16.2% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: Europe
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Switzerland, Japan, United Kingdom
- Emerging Countries: China, India, South Korea, Brazil, Mexico
Last updated on : 25 February, 2026
Biosimilars Market - Growth Drivers and Challenges
Growth Drivers
- Upsurge in the Approval Rates of Biosimilars – which now many other countries are involved in, as biosimilars are safe and effective medications for treating many illnesses and chronic conditions. The surge in the approval of these drugs worldwide is expected to drive the global biosimilar market in the forecasted period. For instance, it was found that 9 new biosimilars were approved in the European Union in 2020 and 7 were approved in 2021.
- Growing Number of Cancer Cases – One of the biggest causes of mortality in the world is cancer, and it is predicted that there will be an increase in the number of new cancer cases. The rise in cancer incidence is driving up demand for biological medications, which are frequently used in cancer therapy. It was found that there were approximately 19 million new cancer cases registered worldwide in 2020.
- Rising Prevalence of Crohn’s disease – The global market for biosimilars is expanding owing to the increased frequency of Crohn's disease. Inflammation of the digestive system is brought on by the inflammatory bowel disorder (IBD) known as Crohn's disease. Although the precise etiology of Crohn's disease is unknown, and it is thought to be the result of a confluence of the immune system, environmental, and genetic variables. As per the data, in North America and Western Europe, 100 to 300 per 100,000 people suffer from Crohn’s disease every year.
- Increasing Number of People Suffering from Kidney Disease – End-stage renal disease (ESRD), which necessitates dialysis or a kidney transplant, can develop from kidney disease. Moreover, Erythropoiesis-stimulating agents (ESAs), among other biologic medications, are frequently used to treat anemia in ESRD patients. The high cost of these medications, however, makes biosimilars a desirable substitute for both patients and healthcare professionals. It was estimated that worldwide over 850 million people were suffering from some form of kidney disease in 2021.
- Rising Cases of Arthritis – Inflammation in the joints is a typical symptom of arthritis, which affects millions of people worldwide. An autoimmune condition called rheumatoid arthritis (RA) damages and inflames the joints, resulting in pain and incapacity. Therefore, the escalating ratio of RA is boosting the requirement of rheumatoid arthritis treatment, which in turn estimated to drive the demand for biological medications. It is observed that in 2021 about 350 million people had arthritis globally.
Challenges
- Increasing Competition in the Market - There is intense competition in the market for both biosimilar manufacturers and originator biologic manufacturers. When new biosimilar products enter the market, the originator manufacturer may use a variety of strategies to compete with the competition, such as competitive pricing, launching second-generation products, reformulations, and improved dosing measures. Hence, such a factor is projected to impede market growth in the coming years.
- Manufacturing Process Concerns for Biosimilar Agents
- The difficulty of Generating an Exact Copy of Biosimilars and Biologics
Biosimilars Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
16.2% |
|
Base Year Market Size (2025) |
USD 36.08 billion |
|
Forecast Year Market Size (2035) |
USD 161.93 billion |
|
Regional Scope |
|
Biosimilars Market Segmentation:
Product Type Segment Analysis
The recombinant non-glycosylated proteins segment is poised to garner highest biosimilars market share by 2035, backed by growing concern of rising incidences of diabetes along with surge in the spread of diabetes worldwide. Furthermore, recombinant non-glycosylated proteins are used for the treatment of the diabetes. For instance, it was estimated that 1.5 million deaths were directly caused by diabetes worldwide, it was the 9th leading cause of death in 2019.
Application Segment Analysis
The oncology segment will account for largest biosimilars market share by 2035, owing to rising number of cancer cases across the globe. Moreover, biosimilars have been created and approved to deliver efficient results in breast cancer treatment, colorectal cancer treatment, and other solid tumor treatments. Chemotherapy and other cancer therapies are combined with biosimilars to increase cancer patient chances of survival and quality of life.
Our in-depth analysis of the global biosimilars market includes the following segments:
|
By Product Type |
|
|
By Technology |
|
|
By Application |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Biosimilars Market - Regional Analysis
Europe Market Insights
Europe biosimilars market is estimated to account for largest revenue share by the end of 2035. The growth of the market can be attributed to presence of a strong healthcare network in the region, and the growing number of hospitals and diseases. As per the reported data, nearly 10 percent of the total population in the United States were suffering from diabetes in the year 2021. Moreover, the rising awareness of biosimilars amongst the healthcare service providers, along with surge in prescribing biosimilars, is anticipated to drive the market growth. Furthermore, the market revenue is propeled by volume of biosimilar prescribing has generated high savings from biosimilar competition.
Biosimilars Market Players:
- Biocon Limited
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Amgen Inc.
- Novartis AG
- Intas Pharmaceuticals Ltd.
- BioXpress Therapeutics SA
- Genor Biopharma Co. Ltd
- Allergan plc (AbbVie Inc.)
- Coherus BioSciences, Inc.
- Pfizer Inc.
- Reliance Life Sciences Private Limited
Recent Developments
-
Biocon Limited - Abevmy (bBevacizumab) is now available in Canada. It is co-developed by Biocon Biologics (a subsidiary of Biocon Limited) and Viatris and is a biosimilar to Roche’s Avastin (Bevacizumab). Health Canada has approved the drug for four oncology indications.
-
Amgen Inc., - announced the approval of RIABNI (rituximab-arrx), a biosimilar to Rituxan (rituximab) by the U.S. Food and Drug Administration (FDA). It is used for the treatment of adult patients with Non-Hodgkin's Lymphoma (NHL), Chronic Lymphocytic Leukemia (CLL), Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis), and Microscopic Polyangiitis (MPA).
- Report ID: 1376
- Published Date: Feb 25, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Biosimilars Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




