Immunoglobulin Replacement Therapy Market Outlook:
Immunoglobulin (IgG) Replacement Therapy Market size was valued at USD 14.31 billion in 2025 and is set to exceed USD 29.22 billion by 2035, registering over 7.4% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of immunoglobulin replacement therapy is estimated at USD 15.26 billion.
The market for the therapy has increased because of the rising number of patients with auto-immune diseases such as IVIG and Guillain-Barré Syndrome, as well as the clinical effectiveness of immunoglobulin in treating these problems. Hence, the increasing frequency of these diseases will escalate the growth of the market. According to a report every year, about 100,000 people globally contract GBS. Also, the predicted incidence of GBS in 2020 was 1.41 cases per 100,000 people a year, up from 0.89 cases in 2019. In 2021, the overall rate of GBS rose by 59%.
Furthermore, the market is driven by the rising incidence of immunodeficiency illnesses. Moreover, the increased prevalence of lifestyle-related issues like obesity and antibody deficiency illnesses are also propelling the market growth. Since immunoglobulin (IgG) replacement therapy is the only viable and effective treatment option currently on the market, demand for it is rising. The immunoglobulin (IgG) replacement therapy market is anticipated to expand quickly throughout the projected period.
Key Immunoglobulin (IgG) Replacement Therapy Market Insights Summary:
Regional Insights:
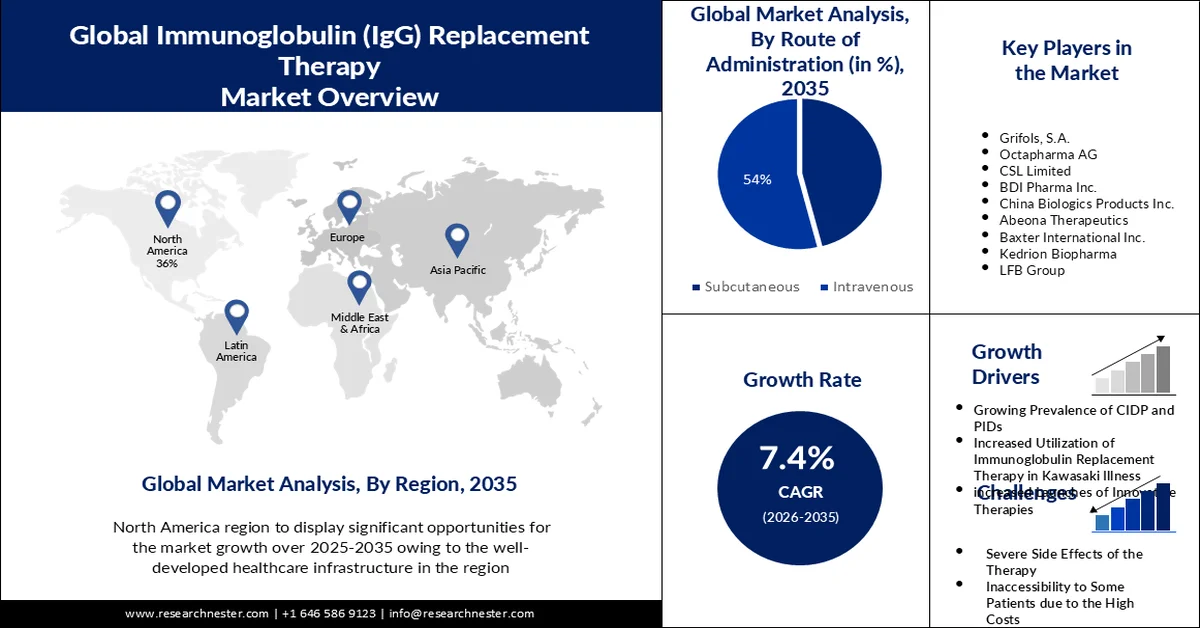
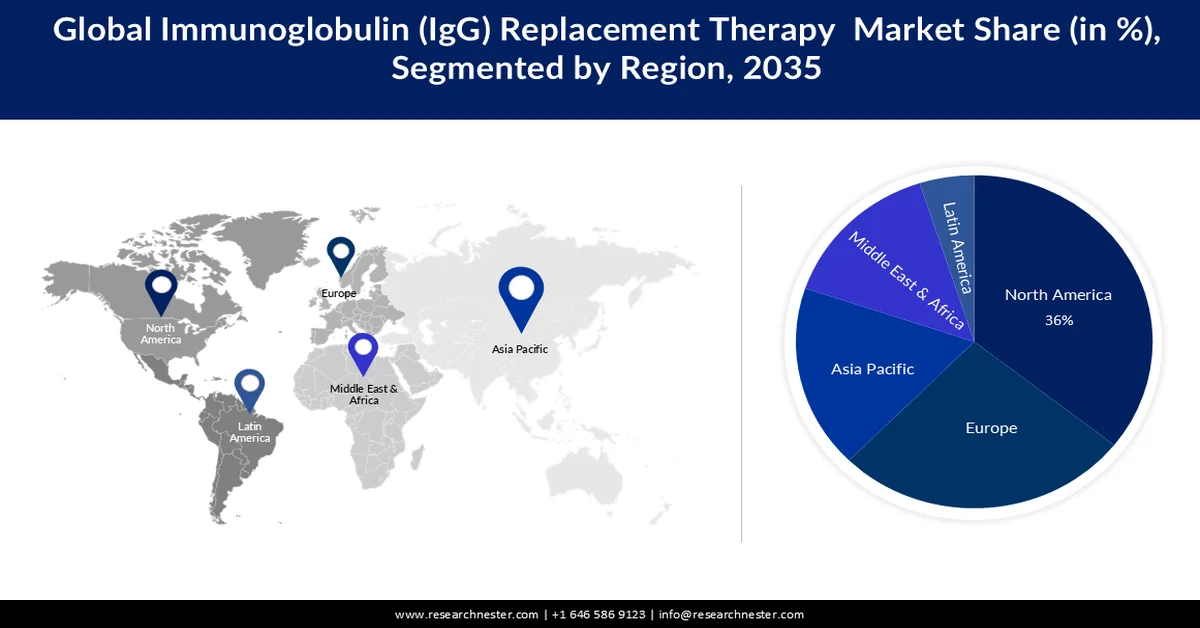
- North America is projected to command a 36% share of the immunoglobulin (igg) replacement therapy market by 2035, propelled by well-developed healthcare infrastructure and the strong presence of major industry players.
- Europe is anticipated to capture 27% share by 2035, fueled by rising incidence of immune-deficiency disorders and increasing physician inclination toward immune-deficiency treatments.
Segment Insights:
- The Intravenous segment of the immunoglobulin (igg) replacement therapy market is set to account for 54% share during the forecast period 2026–2035, driven by rapid immunoglobulin absorption, higher dosage accuracy, and reduced treatment frequency.
- Hypogammaglobulinemia segment is expected to hold 30% share over 2026–2035, owing to growing disease awareness, expanding elderly population, and advancements in diagnostic techniques.
Key Growth Trends:
- Growing Prevalence of CIDP and PIDs Globally
- Increased Utilization of Immunoglobulin Replacement Therapy in Kawasaki Illness
Major Challenges:
- Severe Side Effects of the Therapy
- Inaccessibility to Some Patients due to the High Costs of the Therapy may Hamper the Market Growth
Key Players: Albemarle Corporation, Neogen Chemical Ltd, Chevron Phillips Chemical Company LLC, MATHESON TRI-GAS, Verni Gas Corporation, TOSOH CORPORATION, Tata Chemicals Ltd, Triveni Interchem Private Limited, Sontara Organo Industries, Air Liquide, Showa Denko K.K., ADEKA CORPORATION, Japan Finechem Company, Inc., Kishida Chemical Co., Ltd.
Global Immunoglobulin (IgG) Replacement Therapy Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 14.31 billion
- 2026 Market Size: USD 15.26 billion
- Projected Market Size: USD 29.22 billion by 2035
- Growth Forecasts: 7.4% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (36% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, China, France
- Emerging Countries: China, India, Japan, South Korea, Brazil
Last updated on : 25 February, 2026
Immunoglobulin Replacement Therapy Market - Growth Drivers and Challenges
Growth Drivers
-
Growing Prevalence of CIDP and PIDs Globally - The global market for IgG replacement therapy is expanding quickly as a result of the increasing prevalence of bleeding disorders and CIDP (Chronic Inflammatory Demyelinating Polyneuropathy) in patients. Also, the growing number of people with autoimmune diseases and other pathological problems is driving up demand for IgG replacement therapy solutions. According to a recent study, some of the more than 80 distinct autoimmune illnesses that impact about 4% of the global population are type 1 diabetes, multiple sclerosis, rheumatoid arthritis, lupus, Crohn's disease, psoriasis, and scleroderma. Moreover, the growing incidences of PIDs globally are also propelling the market growth. Many of these immune system abnormalities are brought on by issues with the genetic code, which serves as a blueprint for creating the body's cells (DNA). Primary immunodeficiency illnesses come in over 300 different forms, and the list keeps growing as new ones are found by researchers.
- Increased Utilization of Immunoglobulin Replacement Therapy in Kawasaki Illness - The market is expected to increase commercially due to the rising incidence of Kawasaki illness in children and the increasing need for immunoglobulin replacement therapy as a treatment. Over the coming years, there will likely be more usage of steroids to treat Kawasaki sickness because they are the only therapeutic alternative available (except for immunoglobulin replacement therapy), which has considerable side effects. Furthermore, the demand for immunoglobulin replacement therapy is expected to rise due to factors like elevated patient awareness and a rise in the occurrence of illnesses with insufficient antibody supplies, which will propel the market.
- Increased Launches of Innovative Therapies by the Key Manufacturers - The growing amount of FDA approvals could be a major factor in this industry's growth. For instance, in January 2022, Octapharma declared that a broader spectrum of patients with acquired immune deficiencies now have more flexible treatment options with the expansion of the indication for cutaquig®, a human immunoglobulin for subcutaneous administration, in the European Union (EU). Adults, children, and adolescents with severe or recurring infections, inadequate antimicrobial therapy, and either proven specific antibody failure or a serum immunoglobulin (IgG) level below 4 g/L are included in the expanded cutaquig® prescription. Consequently, several domestic and foreign companies are investing in the IgG replacement therapy market.
Challenges
- Severe Side Effects of the Therapy – Approximately 25% of people may experience adverse effects when taking their medicine. They are not dangerous; however, they are usually bothersome or mild. Headache, fever, chills, exhaustion, sore muscles or joints, and allergic responses are a few typical adverse effects. Usually, these symptoms are transient and mild. More severe side effects, such as allergic reactions, kidney issues, and blood clots, are uncommon but possible. Therefore, the severe side effects of the therapy may hinder the growth of the immunoglobulin (IgG) replacement therapy market.
- Inaccessibility to Some Patients due to the High Costs of the Therapy may Hamper the Market Growth
- Limited Availability of Immunoglobulin Products may Impede the Market Growth
Immunoglobulin Replacement Therapy Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
7.4% |
|
Base Year Market Size (2025) |
USD 14.31 billion |
|
Forecast Year Market Size (2035) |
USD 29.22 billion |
|
Regional Scope |
|
Immunoglobulin Replacement Therapy Market Segmentation:
Route of Administration Segment Analysis
Intravenous segment of the immunoglobulin replacement therapy market is poised to hold the largest share of 54% during the forecast period. Immunoglobulin is infused directly into the bloodstream through a vein during intravenous therapy. This technique makes it possible to swiftly and effectively provide immunoglobulin at a higher concentration, supporting the immune system right away. IV treatment has the advantage of being given less frequently than subcutaneous injections. As a result, patients might need fewer treatment sessions, which would make therapy more convenient and less taxing. Furthermore, patients who need larger doses of immunoglobulin or who have trouble self-administering subcutaneous injections frequently choose intravenous therapy. It guarantees that the immunoglobulin is quickly absorbed by the body and permits accurate dosage. Therefore, these advantages are accelerating the growth of the intravenous segment.
Application Segment Analysis
Immunoglobulin (IgG) Replacement Therapy Market in hypogammaglobulinemia is anticipated to hold a share of 30% during the projected period. The rising awareness and identification of this ailment is the reason for the segment's expansion. An increasing number of individuals are receiving accurate diagnosis and treatment as more medical professionals become informed about the illness and its symptoms. Moreover, the growing senior population, as those over 60 frequently experience poor immunity disorders that interfere with their ability to function normally is also growing the growth of the segment. For instance, the percentage of people over 60 in the world will almost triple from 12% to 22% between 2015 and 2050. Furthermore, the development of diagnostic methods is another aspect driving the segment's expansion. It is now simpler to detect people with hypogammaglobulinemia due to improved screening techniques and laboratory testing, which has allowed for early intervention and therapy. The industry has also grown as a result of the creation of innovative medicines and treatment choices.
Our in-depth analysis of the global immunoglobulin (IgG) replacement therapy market includes the following segments:
|
Route of Administration |
|
|
Application |
|
|
Distribution Channel |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Immunoglobulin Replacement Therapy Market - Regional Analysis
North American Market Insights
North America industry is expected to hold largest revenue share of 36% by 2035. The market is expanding in the region due to the well-developed healthcare infrastructure and a notable prevalence of diseases requiring immunoglobulin therapies. The established healthcare framework enables the delivery of immunoglobulin therapies, contributing to the region’s robust market position. Also, the presence of major key players in the region is propelling the growth of the market in the region. For instance, Pfizer Inc. announced the supplemental biologics license application (sBLA) for PANZYGA® (Immune Globulin Intravenous [Human] – is 10% Liquid Preparation) has been approved by the U.S. Food and Drug Administration (FDA) for the treatment of adult patients suffering from chronic inflammatory demyelinating polyneuropathy (CIDP). Therefore, these factors are propelling the growth of the market in the region.
European Market Insights
Immunoglobulin (IgG) Replacement Therapy Market in Europe is poised to hold the second-largest share of 27% by the end of 2035. Rising incidence of several immune-deficiency diseases and rising physician propensity toward immune-deficiency treatments. Additionally, the market is expanding more quickly because of the region's rising congenital AIDS prevalence. As per a report, of the 53 countries in Europe, 49 reported 110,486 HIV cases in 2022. During this time, the number of deaths among those with an AIDS diagnosis fell, reaching 767 deaths in 2022. Furthermore, immunoglobulin products are now safer and easier for patients to obtain thanks to improvements in production and purifying processes. Since more people can now access the required therapy, this has helped the market expand.
Immunoglobulin Replacement Therapy Market Players:
- Boehringer Ingelheim
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Grifols, S.A.
- Octapharma AG
- CSL Limited
- BDI Pharma Inc.
- China Biologics Products Inc.
- Abeona Therapeutics
- Baxter International Inc.
- Kedrion Biopharma
- LFB Group
Recent Developments
- Boehringer Ingelheim declared that it has begun the Phase 1 research (NCT05658107) to evaluate the safety, tolerability, and pharmacokinetics of BI 765423, its first-in-class IL-11 inhibitor antibody, in healthy volunteers. Boehringer Ingelheim is already a world leader in the management of pulmonary fibrosis, and the company's long-term dedication to treating fibrotic disease across therapeutic domains is demonstrated by the start of the anti-IL-11 treatment's clinical development.
- Leading manufacturer of plasma medications, Grifols, has begun selling its 20% subcutaneous immunoglobulin, XEMBIFY®, in Spain. Primary immunodeficiencies (PID) and some secondary immunodeficiencies (SID), the prevalence of which is predicted to rise sharply in the next several years, are the conditions for which this plasma medication is advised.
- Report ID: 5635
- Published Date: Feb 25, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Immunoglobulin (IgG) Replacement Therapy Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




