Introduction to Haemophilus Influenzae Type B Vaccine
The vaccination against haemophilus influenzae type B started during the 1970s when the first capsular polysaccharide vaccine consisting of polyribosylribitol phosphate was developed. Initially, the studies related to these vaccines remained confined to United States of America and Canada. During the 1980s, modified and conjugated vaccines became commercially available with four different conjugations depending on the type of influenza.
The haemophilus Influenzae Type B is a major cause of mortality and morbidity in young childhood with six of its entities being- meningitis, bacteremic pneumonia, epiglottitis, septicemia, cellulitis, and osteoarticular infections. The HIB disease is the cause of around 90% children deaths per year. The currently available HIB vaccine is highly effective and safe with 99% children developing antibodies, once the course gets completed. The vaccines have been successful in ensuring decline of the disease by up to 80% in the past five years globally.
Market Size and Forecast
The global haemophilus influenzae type B vaccine market is anticipated to record a significant growth over the forecast period i.e. 2019-2027 owing to increase in healthcare initiatives and rise in diseases among infants. Recently, there has been considerable access to these vaccines globally with increased investment of private sector in healthcare which is anticipated to increase the growth of the market. The increasing number of HIB cases worldwide have made these vaccines a regularized immunization program, resulting in rise in demand for the product in both developed as well as developing countries. The recent research and developments taking place in the healthcare sector is expected to improve the availability of vaccines, owing to which the market is anticipated to observe significant growth. The availability of variety in the dosage preparation and different formulations depending on the type of influenza is anticipated to increase the market growth. The HIB vaccines are effective and safe for use by infants, as they contain polyribosylribitol phosphate combined with proteins that is driving their usage in several cases. The rise in cases of HIB diseases for instance, meningitis, pneumonia, epiglottis and others in the children is anticipated to increase the demand for these vaccines and is estimated to drive the growth of the market over the forecast period. Meningitis is most commonly observed in those children suffering from HIB disease, with almost 60% of these cases recorded according to WHO.
Get more information on this report: Download Sample PDF
The market is segmented by type, formulation & application. Formulation segment is divided into liquid monovalent
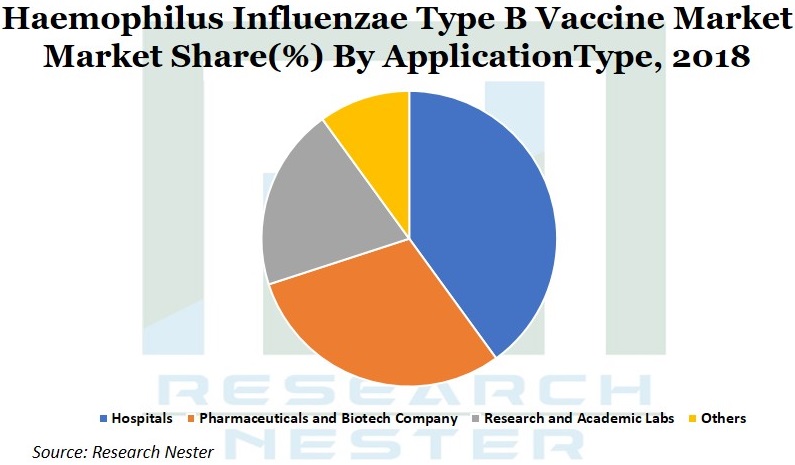
HIB, liquid combination HIB, lyophilized monovalent HIB and lyophilized combination HIB out of which, the liquid combination HIB segment is anticipated to hold the leading share, on account of the fact that no re-constitution is required, multi-dose vials is possible that results in reduced wastage and requires less cold chain space as compared to two monovalent vaccine formulations. The application segment is further segmented into hospitals, research & academic labs, pharmaceuticals and biotechnology companies and others. Out of these segments, the hospitals segment is anticipated to occupy the leading market share, owing to increased cases of HIB recorded, coupled with government regulations on regular immunization programs that are to be followed by pharmaceuticals and biotechnology companies and research & academic labs . On the basis of type, the market is segmented into HIB Vaccine and DTaP-IPV, out of which the DTaP-IPV vaccine is anticipated to hold leading market share, owing to its use in protection of babies starting from 2 months to 2 years old against HIB disease.
Growth Drivers
Initiatives and Investments
The various initiatives by public and private agencies, for instance GAVI coupled with WHO initiative to introduce the haemophilus influenzae type B disease into global immunization programs is anticipated to drive the growth of the market. Owing to the GAVI initiative, 80% of the GAVI covered countries have been successfully provided with vaccines. These initiatives help to provide vaccine to the low income countries at a subsidized rate, on account of which these vaccines are made available at the global level. The various initiatives taken up by companies in the healthcare sector to provide better medicines coupled with constant research undertaken is anticipated to contribute to the growth of the market over the forecast period.
Burden of Disease
The HIB disease may lead to the occurrence of at least 2,00,000 deaths every year, if not vaccinated properly. Moreover, , the disease can also cause further illness leading to paralysis, deafness, hallucinations and many more when left untreated. Owing to this factor, proper vaccination is being encouraged globally that is anticipated to drive the growth of the HIB vaccine market over the forecast period.
Restraints
High Cost and Limited Shelf life
The high vaccine cost, for instance DTap-IP-HI being available at $96 per vaccine and others ranging from $50- $200 per vaccine is anticipated to act as a major restraint to the growth of the market, as low income countries will not be able to bear such high unaffordable costs. The countries who are not part of the international initiatives were unable to buy these HIB vaccines, due to which new antibiotics have been developed that can be used in place of HIB vaccines Additionally, these vaccines possess a limited shelf life, coupled with specific storage conditions for storing these liquid monovalent HIB vaccines that cannot be allowed to freeze, but also at the same time should be stored in temperatures ranging from 2ËšC to 4ËšC. At the same time, patients may have allergy to certain chemicals that is anticipated to reduce the demand and is estimated to hinder the growth of the market over the forecast period.
Market Segmentation
Our-in depth analysis of the haemophilus influenzae type B vaccine market includes the following segments:
By Type
- Type 1-HIB Vaccine
- Type 2- DTaP-IPV
By Formulation
- Liquid Monovalent HIB
- Liquid Combination HIB
- Lyophilized Monovalent HIB
- Lyophilized Combination HIB
By Application
- Hospital
- Research and Academic Labs
- Pharmaceuticals and Biotechnology Companies
- Others
By Region
On the basis of regional analysis, the haemophilus influenzae type B vaccine market is segmented into five major regions including North America, Europe, Asia Pacific, Latin America and Middle East & Africa region.
The market in North America is anticipated to occupy the leading market share, owing to high investment in the healthcare sector and continuous research & development taking place in the region. The regular immunization program initiated by the government is anticipated to increase the market growth in the region. The market in Europe is anticipated to grow significantly over the forecast period, owing to high adoption of vaccines in the region. 51 of 53 member countries have successfully implemented the recommendations of WHO to count HIB as part of their infant immunization program. The market in Asia-Pacific region is anticipated to grow significantly, owing to increased awareness and rise in adoption of vaccines by the population. The region has accepted the WHO recommendation and are also getting associated with GAVI. Some of the countries, for instance are getting involved in developing vaccines which is anticipated to drive the growth of the market over the forecast period. The market in Middle East and Africa region is anticipated to witness decent growth, owing to different international initiatives being undertaken, growing awareness among the population and improved medical facilities.
The haemophilus influenzae type B vaccine market is further classified on the basis of region as follows:
- North America (U.S. & Canada) Market size, Y-O-Y growth & Opportunity Analysis
- Latin America (Brazil, Mexico, Argentina, Rest of Latin America) Market size, Y-O-Y growth & Opportunity Analysis
- Europe (U.K., Germany, France, Italy, Spain, Hungary, Belgium, Netherlands & Luxembourg, NORDIC, Poland, Turkey, Russia, Rest of Europe) Market size, Y-O-Y growth & Opportunity Analysis
- Asia-Pacific (China, India, Japan, South Korea, Indonesia, Malaysia, Australia, New Zealand, Rest of Asia-Pacific) Market size, Y-O-Y growth & Opportunity Analysis.
- Middle East and Africa (Israel, GCC (Saudi Arabia, UAE, Bahrain, Kuwait, Qatar, Oman), North Africa, South Africa, Rest of Middle East and Africa) Market size, Y-O-Y growth & Opportunity Analysis
Top Featured Companies Dominating the Market
- Novartis
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- Merck Sharp & Dohme Corp
- Sanofi Pasteur
- GlaxoSmithKline
- Takeda Pharmaceuticals Company Ltd.
- Walvax Biotechnology Co. Ltd.
- Lanzhou Institute of Biological Products Co., Ltd
- Report ID: 1947
- Published Date: Feb 14, 2023
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert





